First non-human primate study showing promise of gene therapy for stroke repair
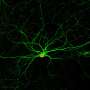
Stroke is a leading cause of death and severe long-term disability with limited treatments available. A research team led by Prof. Gong Chen at Jinan University, Guangzhou, China recently reported the first non-human primate study demonstrating successful in vivo neural regeneration from brain internal glial cells for stroke repair. This work was published in Frontiers in Cell and Developmental Biology.
"Current treatment of ischemic stroke mainly aims at restoring blood flow and neuroprotection, typically with a narrow time window of several hours. However, many stroke patients cannot reach hospital within a few hours after the onset of stroke, and may suffer from a large number of neuronal death and loss of brain functions. Therefore, how to regenerate functional new neurons after stroke is the key to restore brain functions," said Long-Jiao Ge, the first author of the work and a Ph.D. student in the Institute of Zoology, Chinese Academy of Sciences.
"We have previously published a series of works demonstrating that overexpression of a single neural transcriptional factor NeuroD1 can directly convert glial cells into neurons in mouse brains. However, most clinical trials on stroke have failed in the past decades, suggesting that successful rodent studies may not be sufficient for clinical translation. Because in vivo glia-to-neuron conversion is such an innovative technology, we decided to take an important step to further validate this new technology in non-human primates," she said, describing the original intention of this work.
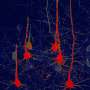
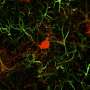
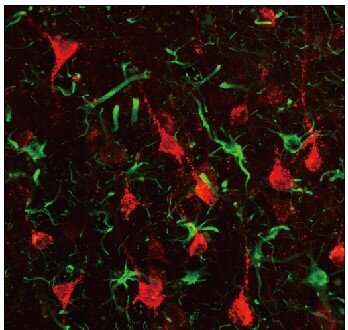
In this study, Prof. Chen and his team first established an ischemic stroke model in rhesus macaque monkeys aged from 9 to 21 years old to capture the typical occurrence of stroke among senior population of humans. Using astrocyte promoter GFAP to control the expression of neural transcriptional factor NeuroD1, Chen's team demonstrated successfully that reactive astrocytes caused by ischemic stroke in the monkey brain can be efficiently converted into neurons. "We are very happy to see that the neuronal density in NeuroD1-treated areas is consistently higher than that not treated with NeuroD1," said Ge.
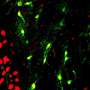
"An unexpected finding is that a class of interneurons, that are sensitive to stroke injury, are significantly protected after NeuroD1 treatment, accompanied by a significant reduction of neuroinflammation. This result has important implication that in vivo astrocyte-to-neuron conversion not only regenerates new neurons but also protects the injured neurons from secondary damage, preventing further neuronal loss. Such findings of combinatorial effects of neuroregeneration plus neuroprotection may have profound impact on brain repair," said Prof. Chen.
"What we have developed here is a unique gene therapy, using adeno-associated virus (AAV) vectors to deliver transgene NeuroD1 through direct intracranial injection into the ischemic region in primate brains. Different from classical gene therapies aiming for correction of gene mutations, our gene therapy regenerates new neurons, making it a kind of gene therapy-mediated cell therapy. We call it neuroregenerative gene therapy," Prof. Chen said.
"This study in non-human primates opens a new avenue using neuroregenerative gene therapy to repair damaged brains, which brings new hope to millions of patients suffering from stroke and other neurological disorders," concluded Prof. Chen.
More information: Long-Jiao Ge et al, In vivo Neuroregeneration to Treat Ischemic Stroke Through NeuroD1 AAV-Based Gene Therapy in Adult Non-human Primates, Frontiers in Cell and Developmental Biology (2020). DOI: 10.3389/fcell.2020.590008