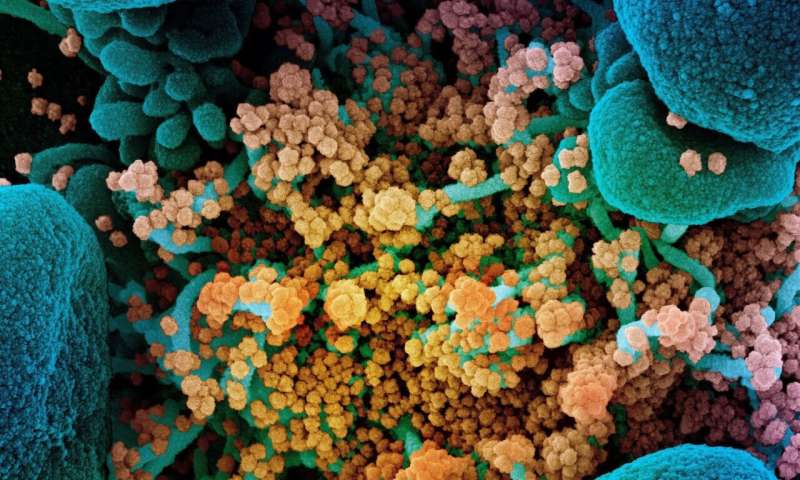
Colorized scanning electron micrograph of a dying cell (blue) heavily infected with SARS-CoV-2 (yellow), the virus that causes COVID-19. Credit: NIAID Integrated Research Facility, Fort Detrick, Maryland.
Real-world disease and parasite monitoring is often hampered by the inability of traditional approaches to easily sample broad geographical areas and large numbers of individuals. This can result in patchy data that fall short of what researchers need to anticipate and address outbreaks. Writing in BioScience, Jessica Farrell (University of Florida), Liam Whitmore (University of Limerick), and David Duffy (University of Florida) describe the promise of novel molecular techniques to overcome these shortcomings.
By sampling environmental DNA and RNA (eDNA and eRNA), the authors say, researchers will be better able to determine the presence of both human and wildlife pathogens. The eDNA and eRNA approach works through the collection of a sample (often from an aquatic source), whose genetic contents are then sequenced to reveal the presence and prevalence of pathogens. This eDNA or eRNA gives researchers a timely view into disease spread, which "can help predict the spread of pathogens to nearby new and susceptible geographic locations and populations in advance, providing opportunities to implement prevention and mitigation strategies," say the authors.
For instance, during the COVID-19 pandemic, researchers have used eRNA analysis of wastewater to track large-scale outbreaks of disease, finding that "wastewater detection of SARS-CoV-2 eRNA increased rapidly prior to medical detection of human outbreaks in those regions, with environmental virus concentration peaking at the same time or before the number of human-detected cases, providing advanced warning of a surge in infected individuals." With this advance knowledge, crucial and limited medical resources can be provisioned where they will be most needed.
The benefits of eDNA and eRNA analysis are not limited to the detection of human pathogens; the authors describe the ways in which these tools also help in understanding the presence and transmission of pathogens that hamper wildlife conservation efforts, such as the turtle-specific DNA virus, chelonid herpesvirus 5. eDNA monitoring of this pathogen may help researchers evaluate the disease's spread—in particular, the idea that the virus is most frequently transmitted by "superspreader" individuals.
The future for these technologies is bright, say Farrell, Whitmore, and Duffy, "with the potential to vastly exceed traditional detection methods and the capacity to improve the detection and monitoring of aquatic pathogens and their vulnerable host species, including humans."
More information: BioScience (2020). DOI: 10.1093/biosci/biaa158
Journal information: BioScience
Provided by American Institute of Biological Sciences