Newly developed COVID vaccine could protect against omicron and other variants
The preclinical data for a vaccine developed at MedUni Vienna to protect against SARS-CoV-2 indicates that it is effective against all SARS-CoV-2 variants known to date, including omicron—even in those who have not yet built up any immunity as a result of vaccination (non-responders). The data from the study were recently published in the journal Allergy.
The antigen-based vaccine developed at MedUni Vienna, under the leadership of Rudolf Valenta from the Center for Pathophysiology, Infectiology and Immunology, targets the receptor binding domains (RBD) of the SARS-CoV-2 virus and induced a robust and uniform RBD-specific IgG antibody response in animal models and in human tests. This antibody response prevents the virus from docking onto and entering the body's cells, so that infection cannot occur.
Combination of coronavirus vaccine and hepatitis B vaccine
The SARS-CoV-2 subunit vaccine (PreS-RBD) developed at MedUni Vienna is based on a structurally folded fusion protein consisting of two receptor binding domains (RBD) of the SARS-CoV-2 virus and the PreS antigen from hepatitis B, which serve as immunological carriers for each other, thereby strengthening the immune response. Currently available genetic SARS-CoV-2 vaccines induce mainly transient IgG1 antibody responses, whereas the PreS-RBD vaccine can additionally induce long-lived RBD-specific IgG4 antibodies.
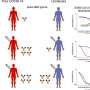
PreS-RBD-specific IgG antibodies detected in blood and mucosal secretions reacted with SARS-CoV-2 variants, including the omicron variant. Antibodies induced by vaccination with PreS-RBD more potently inhibited the binding of RBD with its human receptor ACE2, and their virus-neutralizing titers were higher than those in a random sample of individuals fully immunized with two vaccinations of currently registered vaccines or than those of COVID-19 convalescents (i.e., individuals who had previously had COVID-19).
Immunity even for previous 'non-responders'
"The PreS-RBD vaccine has the potential to induce sterilizing immunity to old and new SARS-CoV-2 variants by preventing infection by stopping viral replication and transmission through the inhibition of cellular virus entry," explains study leader Rudolf Valenta. Moreover, it is expected that the vaccine will even be effective in people who have not previously responded to vaccination ("RBD non-responders"), as they will receive additional T-cell support from the PreS portion of the vaccine. An earlier study by Valenta and colleagues had found that approximately 20% of those recovered from COVID-19 failed to form RBD-specific antibodies and were thus at constant risk of re-infection.
Results based on decades of experience from allergy research at MedUni Vienna
The development of this Austrian COVID vaccine was to a large extent inspired by decades of experience in allergy vaccine design. Previous work on allergy vaccines and clinical trials also conducted with PreS-based allergy vaccines have demonstrated the safety of PreS-based vaccines, even when used repeatedly.
"Our data give us grounds to hope that this readily producible protein-based vaccine antigen will be effective against all SARS-CoV-2 variants known to date, including omicron," says study leader Rudolf Valenta. "The vaccine is designed to enable repeated injections to build up sustained sterilizing immunity, is suitable for use in all age and risk groups and appears to be superior to currently available vaccines when it comes to inducing neutralizing antibodies." If sufficient funding is forthcoming, the first clinical trials required for approval could be carried out this year.
More information: Pia Gattinger et al, Vaccine based on folded RBD‐PreS fusion protein with potential to induce sterilizing immunity to SARS‐CoV‐2 variants, Allergy (2022). DOI: 10.1111/all.15305