Establishment of a pancreatic cancer animal model using a hydrodynamic gene delivery method
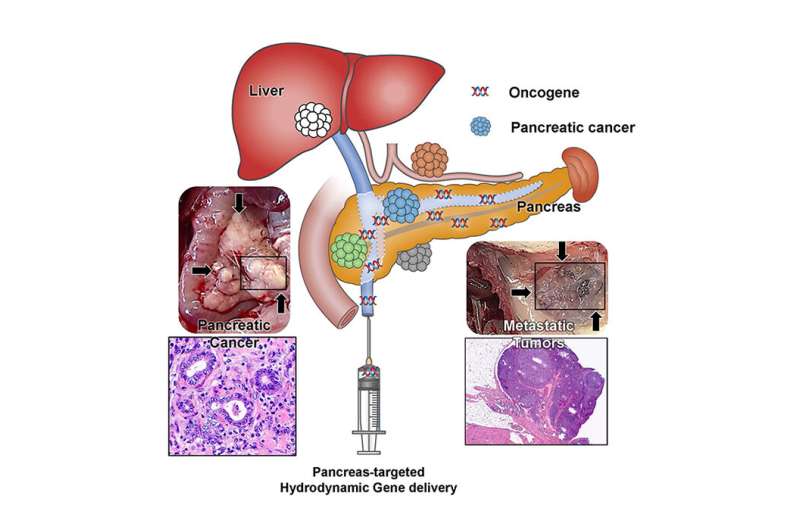
Pancreatic cancer has a significantly poor prognosis; therefore, the development of effective treatments is an unmet clinical need. The major drawback in this field was the lack of useful model animals, which delayed the establishment of markers for early diagnosis and therapeutic options. The research group established an effective carcinogenesis method with wild-type rats by selectively introducing oncogenes into the pancreas, using the pancreas-targeted hydrodynamic gene delivery method that has been developed by the group.
The research group of Professor Kamimura in Niigata University have established a novel pancreatic carcinogenesis model in wild-type rats utilizing the pancreas-targeted selective hydrodynamic gene delivery method developed by their research group.
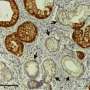
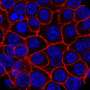
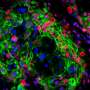
"The gene human pancreatic cancer-related gene transfer of the KRASG12D efficiently developed the pancreatic cancer," says Prof. Kamimura. KRASG12D-induced pancreatic intraepithelial neoplasia lesions showed malignant transformation in the main pancreatic duct at four weeks and developed acinar-to-ductal metaplasia, which led to pancreatic ductal adenocarcinoma within five weeks and the gene combination of KRASG12D and YAP enhanced these effects.
In addition, the combination of oncogenes revealed the metastatic tumors in the liver, lymph nodes, etc., and invasive growth to the surrounding organ and tissues, mimicking the clinical course of human pancreatic cancer. The pancreas-targeted hydrodynamic gene delivery showed its efficacy in developing novel animal models and is promising methods for the organ specific gene therapy. Prof. Kamimura concluded that this pancreatic cancer model will speed up pancreatic cancer research for novel treatments and biomarkers for early diagnosis.
More information: Osamu Shibata et al, Establishment of a pancreatic cancer animal model using the pancreas-targeted hydrodynamic gene delivery method, Molecular Therapy—Nucleic Acids (2022). DOI: 10.1016/j.omtn.2022.03.019