New experimental model for vascular malformations leads to the identification of miransertib as a promising therapy
A team of researchers led by Dr. Sandra Castillo and Dr. Mariona Graupera, from the Josep Carreras Leukaemia Research Institute and CIBERONC, have found the AKT inhibitor miransertib could be a new, effective molecular treatment against low-flow vascular malformations, a long-awaited milestone. Their results, published in the journal EMBO Molecular Medicine, are based on a new in vivo model of blood vessel growth (angiogenesis) that could become the gold standard for this kind of study in the short term.
Vascular malformations are a heterogeneous group of diseases characterized by an abnormal growth of blood vessels with a major impact on patients' quality of life. These alterations happen anywhere in the body and originate mostly during embryonic development. Either as part of a known syndrome or spontaneous, they usually involve mutations leading to the overactivation of PI3K, a key protein in the internal crosstalk of endothelial cells.
Castillo and Graupera discovered in 2016, in collaboration with Dr. Josep Baselga and Dr. Eulàlia Baselga, that vascular malformations were caused by mutations in PI3K. Since then, inhibitors of the PI3K pathway have been the most obvious candidates to treat these diseases, but a poor understanding of the onset and progression of vascular malformations has hindered major advances so far. The reported results, still in the preclinical arena and far from an actual clinical approach, take the first steps to find molecular features to target with specific drugs.
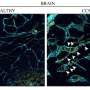
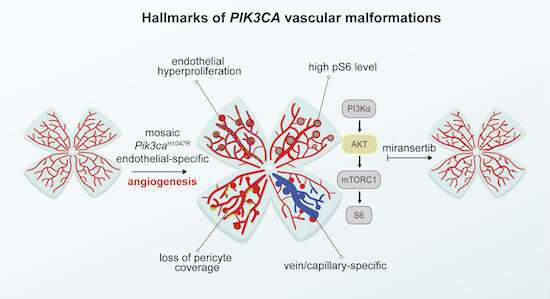
By using a new mouse retina-derived in vivo model, the researchers have been able to mimic the formation of low-flow vascular malformations and determine that, indeed, the PI3K/AKT signaling pathway is the main affected cellular route. According to the research, mutations in the catalytic domain of PI3K seem to trigger a proliferative state in endothelium cells, those lining the inner part of blood vessels, leading to the formation of the characteristic lesions of the disease.
Piotr Kobialka and Helena Sabata, authors of the research, induced the vascular malformations in mutant mice and then treated them with miransertib, an AKT inhibitor used in anticancer therapy. The researchers reported that, in the model, endothelial cells responded to treatment by reducing its proliferation and its capacity to form altered vasculature. These results were further confirmed by using isolated endothelial cells from actual patients, in vitro.
Furthermore, researchers found that active angiogenesis is needed for the malformations to take place, explaining why low-flow alterations are seldomly produced in adulthood, opening the door to preventive strategies in patients already bearing PI3K mutations. In those patients, abnormal blood vessel formation may happen during their life after wound healing or due to hormonal changes.
Overall, the research proposes the first promising candidate for a future molecular therapy on low-flow vascular malformations and offers a fast and robust preclinical model to study angiogenesis in vivo and develop new and more advanced therapies in the short term.
More information: Piotr Kobialka et al, The onset of PI3K‐related vascular malformations occurs during angiogenesis and is prevented by the AKT inhibitor miransertib, EMBO Molecular Medicine (2022). DOI: 10.15252/emmm.202115619