Can the protein that defeats metabolic diseases conquer dementia?
Alzheimer's disease is a representative neurodegenerative disease showing up at an increasing rate in an aging society. Sufferers of this disease are not only portrayed in media, but can often be seen around us. Patients with Alzheimer's disease and their families continue to suffer as the precise cause of dementia remains unknown, and it can only be treated with symptom-relieving therapeutics.
To this end, Professor Kyong-Tai Kim and Ph.D. candidate Eun Ji Oh of the Department of Life Sciences at POSTECH have developed a substance that activates the peroxisome proliferator activated receptor (PPAR), thereby revealing the possibility of treating Alzheimer's disease. PPAR is a transcription factor necessary for metabolic regulation and has been primarily considered as a target protein for the treatment of metabolic diseases.
In patients with Alzheimer's disease, cognitive function and memory gradually decrease as abnormal misfolded proteins form aggregates in brain tissue, and nerve cells are damaged due to chronic inflammatory responses. Recently, Alzheimer's disease has been referred to as type 3 diabetes, and its association with metabolic diseases has been reported.
When insulin resistance increases due to an excessive amount of fat accumulation, metabolism becomes impaired and chronic inflammation in the body increases. When this phenomenon is coupled with aging, the formation of amyloid-β aggregates in brain tissue is accelerated, which damages nerve cells.
Therefore, the research team focused on PPAR, which has been studied as a therapeutic target for several metabolic diseases, such as obesity, dyslipidemia, and diabetes. Through a drug development platform that simultaneously uses virtual computer screening and cell-based screening techniques, the research team successfully developed a small molecule compound and demonstrated that it could activate PPAR by confirming its binding to actual PPAR proteins.
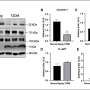
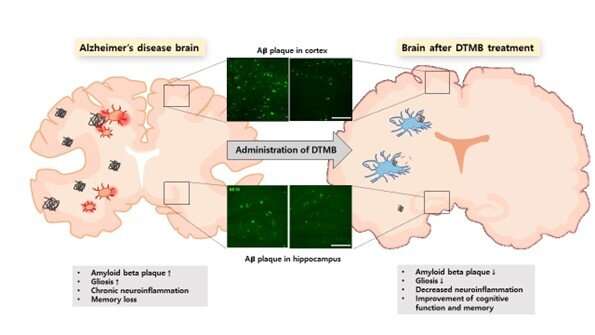
Following the oral administration of this compound for 3 months in mice model with Alzheimer's disease, memory and cognitive function that were degraded due to dementia were found to be restored to the same level as in the normal mouse model. Further, the amount of amyloid-β aggregates and the extent of neurogliosis were reduced.
Additionally, the research team discovered that chronic inflammation caused by amyloid-β peptides in the immune cells of brain tissue was significantly reduced. Professor Tae Young Roh determined that the expression of inflammation-related genes was suppressed and Professor Im-Sook Song in the College of Pharmacy at Kyungpook National University confirmed that the drug penetrated the blood–brain barrier (BBB) and was delivered to the brain tissue.
Professor Kyong-Tai Kim explained that "this research will be of great help to patients suffering from Alzheimer's disease." He added that "in the future, we plan to develop a drug optimized for the treatment of Alzheimer's disease through toxicity testing and structure-activity relationship analysis."
This study was recently published in the journal Neurotherapeutics.
More information: Eunji Oh et al, Synthetic PPAR Agonist DTMB Alleviates Alzheimer's Disease Pathology by Inhibition of Chronic Microglial Inflammation in 5xFAD Mice, Neurotherapeutics (2022). DOI: 10.1007/s13311-022-01275-y