Significant antiviral activity of bulevirtide for treatment of chronic hepatitis D infection
Between 10 and 20 million people worldwide are infected with hepatitis D virus (HDV). HDV infection is always associated with hepatitis B virus infection. So far there has been no satisfactory therapeutic option for this most severe form of chronic viral hepatitis. In a multicenter Phase II clinical trial, an international research team was now able to show that the 24-week application of the drug candidate bulevirtide, developed by DZIF virologist Prof. Stephan Urban, significantly reduced hepatitis D viral load in blood serum and liver. Treatment with the novel entry inhibitor therefore represents a promising strategy for patients with chronic HDV infection.
Of the approximately 250 million people worldwide chronically infected with hepatitis B virus (HBV), between 10 and 20 million are also infected with hepatitis D virus (HDV). HDV uses HBV as a helper virus by packaging its RNA genetic material into HBV envelopes. This co-infection leads to the most severe course of viral liver disease. Unfortunately, the antiviral drugs currently available to treat HBV-infected patients can only be used effectively in a small proportion of HDV patients and may also cause side effects. With the development of bulevirtide—an antiviral agent conditionally approved in the EU since 2020 that blocks the entry of hepatitis B and hepatitis D viruses into liver cells—the goal of successfully treating hepatitis D has come much closer.
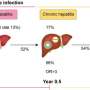
The antiviral activity of bulevirtide was tested in a multicenter Phase II study in a total of 120 HBV/HDV-positive patients, 59 of whom had already developed liver cirrhosis. The analysis of the study, published in the journal The Lancet Infectious Diseases, showed that application of bulevirtide for 24 weeks significantly reduced the concentration of HDV RNA in the blood serum and liver of the subjects and was well tolerated by the study participants.
Although the concentration of hepatitis D virus RNA increased again in most patients after cessation of the medication, the study demonstrates very good response rates, but also indicates that longer-term treatment with bulevirtide is necessary. Efficacy in patients with established cirrhosis continues to demonstrate the safe applicability of bulevirtide in those with advanced liver disease, although no patients with decompensated cirrhosis—a condition denoting end-stage liver disease—were treated. Whether long-term treatment can also lead to permanent reduction of the viral load or even a complete loss of the virus is being investigated in ongoing further studies.
"For affected patients, the study results are of great relevance. Crucially, not only were reduced hepatitis D virus levels observed, but in most cases liver values improved significantly. Furthermore, the daily injections are no problem in everyday life of those affected. The tolerability is really excellent," says DZIF-scientist Prof. Heiner Wedemeyer, director of the Department of Gastroenterology, Hepatology and Endocrinology at Hannover Medical School and clinical leader of the phase II and III studies on bulevirtide.
"Due to the availability of liver biopsies, this study provides unique opportunities to investigate viral load and thus the efficacy of bulevirtide not only serologically, but also in the liver, the site of viral replication," says study co-author and DZIF-scientist Prof. Maura Dandri of the University Medical Center Hamburg-Eppendorf.
"The successful development of bulevirtide from basic research into clinical practice is of drastic importance for many patients for whom there was no treatment option so far," says study last author Prof. Stephan Urban, DZIF-Professor for Translational Virology and head of the Hepatitis B Research Group at Heidelberg University.
More information: Heiner Wedemeyer et al, Safety and efficacy of bulevirtide in combination with tenofovir disoproxil fumarate in patients with hepatitis B virus and hepatitis D virus coinfection (MYR202): a multicentre, randomised, parallel-group, open-label, phase 2 trial, The Lancet Infectious Diseases (2022). DOI: 10.1016/S1473-3099(22)00318-8