Predictive molecular biomarkers for determining neoadjuvant chemosensitivity in muscle invasive bladder cancer
A new research paper was published in Oncotarget entitled "Predictive molecular biomarkers for determining neoadjuvant chemosensitivity in muscle invasive bladder cancer."
Identifying neoadjuvant chemotherapy (NAC) response in patients with muscle invasive bladder cancer (MIBC) has had limited success based on clinicopathological features and molecular subtyping. Identification of chemotherapy responsive cohorts would facilitate delivery to those most likely to benefit.
In this new study, researchers Neal Murphy, Andrew J. Shih, Paras Shah, Oksana Yaskiv, Houman Khalili, Anthony Liew, Annette T. Lee, and Xin-Hua Zhu from Donald and Barbara Zucker School of Medicine at Hofstra/Northwell, Northwell Health Cancer Institute, Feinstein Institutes for Medical Research, and Mayo Clinic aimed to develop a molecular signature that can identify MIBC NAC responders (R) and non-responders (NR) using a cohort of known NAC response phenotypes, and to better understand differences in molecular pathways and subtype classifications between NAC R and NR.
"Therefore, our MIBC patient population with its known chemotherapy response phenotype represents a unique cohort to understand both the molecular mechanisms driving NAC response and to identify a molecular signature that truly correlates with NAC response," say the researchers.
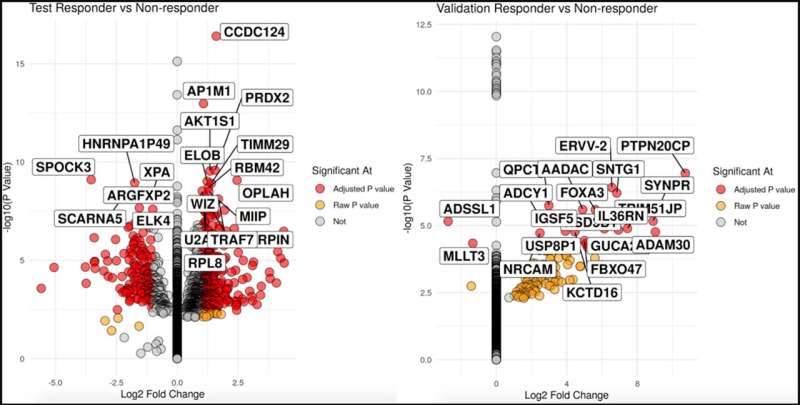
The team presented the messenger RNA (mRNA) and microRNA (miRNA) differential expression profiles from initial transurethral resection of bladder tumor (TURBT) specimens of a discovery cohort of MIBC patients consisting of 7 known NAC R and 11 NR, and a validation cohort consisting of 3 R and 5 NR. Pathological response at time of cystectomy after NAC was used to classify initial TURBT specimens as R (pT0) versus NR (≥pT2). RNA and miRNA from FFPE blocks were sequenced using RNAseq and qPCR, respectively.
The discovery cohort had 2309 genes, while the validation cohort had 602 genes and 13 miRNA differentially expressed between R and NR. Gene set enrichment analysis identified mitochondrial gene expression, DNA replication initiation, DNA unwinding in the R discovery cohort and positive regulation of vascular associated smooth muscle cell proliferation in the NR discovery cohort.
Canonical correlation (CC) analysis was applied to differentiate R versus NR. 3 CCs (CC13, CC16, and CC17) had an AUC >0.65 in the discovery and validation dataset.
Gene ontology enrichment showed CC13 as nucleoside triphosphate metabolic process, CC16 as cell cycle and cellular response to DNA damage, CC17 as DNA packaging complex. All patients were classified using established molecular subtypes: Baylor, UNC, CIT, Lund, MD Anderson, TCGA, and Consensus Class. The MD Anderson p53-like subtype, CIT MC4 subtype and Consensus Class stroma rich subtype had the strongest correlation with a NR phenotype, while no subtype had a strong correlation with the R phenotype.
"Our results identify molecular signatures that can be used to differentiate MIBC NAC R versus NR, salient molecular pathway differences, and highlight the utility of molecular subtyping in relation to NAC response," conclude the researchers.
More information: Neal Murphy et al, Predictive molecular biomarkers for determining neoadjuvant chemosensitivity in muscle invasive bladder cancer, Oncotarget (2022). DOI: 10.18632/oncotarget.28302


















