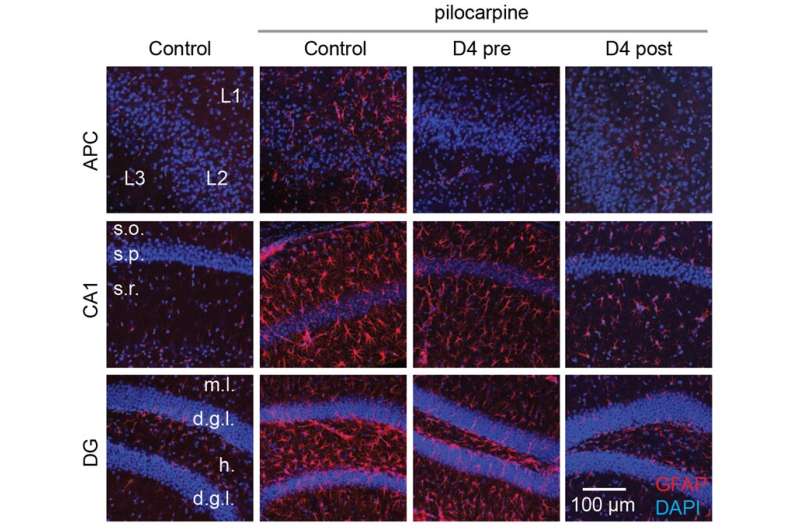
D4 treatment decreases the proliferation and activation of astrocytes in two different brain regions. Pilocarpine was injected into the mice intraperitoneally to induce seizures. Seven days after pilocarpine-induced seizures, the researchers observed a chronic elevation in the number of astrocytes (marked by GFAP staining in red; all cell nuclei marked by DAPI staining in blue). A single injection of the new hemichannel antagonist D4 before or after the induced seizures significantly curbed the proliferation of astrocytes, thus reducing neuroinflammation. Credit: Proceedings of the National Academy of Sciences (2022). DOI: 10.1073/pnas.2213162119
Temporal lobe epilepsy (TLE) is one of the most common types of epilepsy worldwide. Although symptomatic medications are available, one-third of TLE patients remain unresponsive to current treatment, so new drug targets are critically needed. A research team co-led by a City University of Hong Kong (CityU) neuroscientist has recently identified and developed a new drug candidate with the potential for effectively treating TLE by suppressing neuroinflammation.
Epilepsy is one of the most prevalent chronic brain disorders and is characterized by recurrent and spontaneous seizures. Most anti-epileptic drugs that are currently available target neurons and synapses in the brain. They are effective in changing neural circuits and synapses, but this treatment overlooks another important pathology: neuroinflammation.
Neuroinflammation is caused by the abnormal functioning of reactive glial cells, such as astrocytes and microglia, causing an immune reaction in the brain. Accumulating evidence points to a key role of connexin-based gap junctions and hemichannels in brain glial cells in TLE. A hemichannel is a channel or pathway formed by the assembly of six proteins, which permits small molecules such as glutamate to be released from astrocytes and microglia to extracellular space. A gap junction is formed when the hemichannels of two adjacent cells dock with each other.
But inhibiting both gap junctions and hemichannels can lead to undesirable side effects because the former coordinate physiological functions of cell assemblies. Therefore, scientists need to find a way to block only connexin hemichannels to effectively reduce neuroinflammation with fewer side effects.
A research team co-led by Dr. Geoffrey Lau Chun-yue, Assistant Professor in the CityU Department of Neuroscience, identified a new, small organic molecule called D4, which selectively blocks connexin hemichannels, but not gap junctions. The team investigated its effect in treating TLE using a mouse model. The findings suggest that D4 strongly suppresses the TLE-induced neuroinflammation, curbs TLE seizures, and increases the animal's survival rate.
The findings were published in the international scientific journal Proceedings of the National Academy of Sciences (PNAS) under the title "Inhibition of connexin hemichannels alleviates neuroinflammation and hyperexcitability in temporal lobe epilepsy".
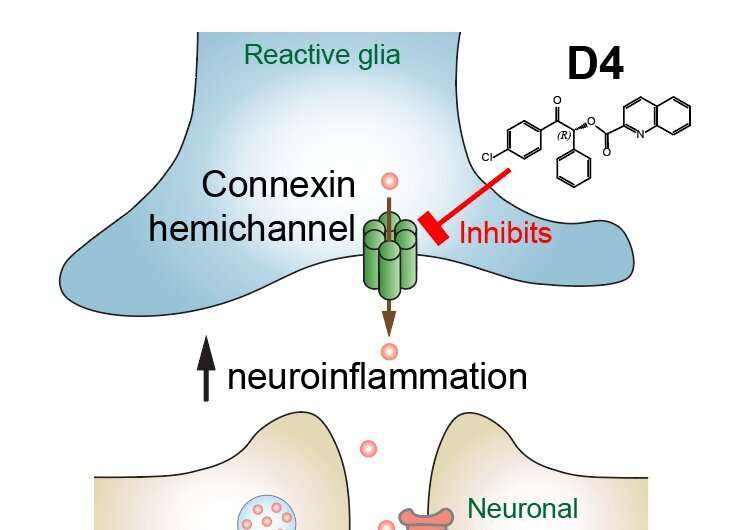
Schematic model of how the new drug, D4, inhibits hemichannels and alleviates neuroinflammation and seizure symptoms. The new drug, D4, is a selective blocker of connexin hemichannels. Excessive glutamate and other molecules can leak out from reactive glia to the extracellular environment, altering synapses, enhancing neuroinflammation and exacerbating seizures. D4 blocks this pathway, suppresses neuroinflammation, and curbs the further development and occurrence of seizures. The latest research findings indicate that D4 is a promising candidate drug for treating epileptic seizures by targeting connexin hemichannels and neuroinflammation. Credit: Dr Geoffrey Lau Chun-yue
New drug D4 suppresses neuroinflammation
"These are very exciting and encouraging results for translational research in epilepsy," said Dr. Lau. "We have found a very promising new drug candidate for treating epilepsy that works through a new mechanism—blocking connexin hemichannels. Our findings also highlight the important involvement of neuroinflammation in neurological disorders such as epilepsy."
The new drug, D4, targets a new class of ion channels, the connexin hemichannels in the glial cells. Glial cells include astrocytes and microglia and are important for modulating neurotransmission. Excessive glutamate and other molecules can leak out from reactive glia via hemichannels to the extracellular environment, altering synapses, enhancing neuroinflammation and exacerbating seizures. By specifically blocking connexin hemichannels using D4, Dr. Lau's team can directly target neuroinflammation caused by astrocytes and microglia.
The research adopted the pilocarpine model of epilepsy in mice, a well-known model to produce phenotypes that resemble human TLE. Pilocarpine was injected into mice intraperitoneally to induce seizures. The administration of one dose of D4 orally before inducing seizures effectively reduced neuroinflammation and altered synaptic inhibition, which increased the animal's survival rate. For treatment after induced seizures, a single dose of D4 had a prolonged effect on suppressing the activation of astrocytes and microglia. This suggests that D4 strongly alleviates neuroinflammation and has a long-term effect.
A single dose offers long-term benefits
Results from both pre- and post-treatment indicate that targeting connexin hemichannels by D4 is an effective and promising strategy for treating epilepsy in which neuroinflammation plays a critical role. The drug can be taken orally to effectively get into the mouse brain to reduce the harmful effects of neuroinflammation. A single dose provides strong protection against future seizures.
"We hope that this will ultimately result in new and better treatment options for epileptic patients," said Dr. Lau. The team will continue to work on the astrocytic mechanisms of epilepsy and identifying more new therapeutic targets.
More information: Anni Guo et al, Inhibition of connexin hemichannels alleviates neuroinflammation and hyperexcitability in temporal lobe epilepsy, Proceedings of the National Academy of Sciences (2022). DOI: 10.1073/pnas.2213162119
Journal information: Proceedings of the National Academy of Sciences
Provided by City University of Hong Kong