First results on the longer-term effects of therapies for the treatment of critically ill patients with COVID-19
The world's largest trial into the effect of multiple interventions for critically ill adults with COVID-19 on longer-term outcomes has released results from the 180-day (six month) follow-up of 4,869 critically ill patients.
Published today in the Journal of the American Medical Association (JAMA), the study is part of the ongoing Randomized Embedded Multifactorial Adaptive Platform for Community Acquired Pneumonia (REMAP-CAP) trial and was led by Monash University's Dr. Lisa Higgins from the School of Public Health and Preventive Medicine.
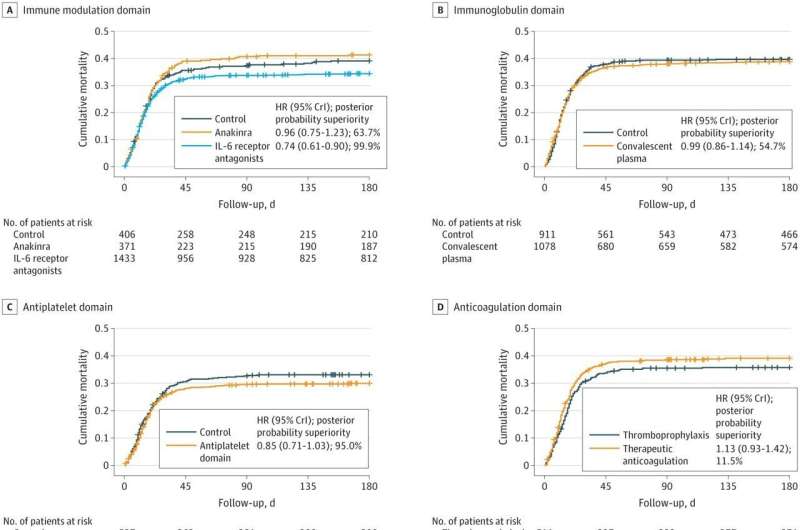
The study found that among critically ill patients with COVID-19 randomized to receive one or more therapeutic interventions, treatment with an IL-6 receptor antagonist (tocilizumab or sarilumab), led to a greater than 99.9% probability of improved 6-month mortality, while antiplatelet agents had a 95% probability of improving 6-month survival.
Moreover, the study also showed that treatments which improve survival, such as interleukin-6 receptor antagonists, do not come at the expense of survival with increased disability or poorer health-related quality of life.
In contrast, longer-term outcomes were not improved with therapeutic anticoagulation, convalescent plasma, or lopinavir-ritonavir, and were worsened with hydroxychloroquine alone or in combination with lopinavir-ritonavir. Corticosteroids did not confer a high probability of improved longer-term survival, although enrolment into this domain was closed early in response to external evidence.
"While we have been treating critically ill patients with COVID-19 for nearly three years, we are still very much in the early stages of understanding the long-term survival, health-related quality of life, and disability among patients with critical illness due to COVID-19," said Dr. Higgins.
"These findings have important clinical and research implications in COVID-19, and in critical care more generally, because they provide evidence that short-term within-hospital treatment effects are sustained longer-term."
The 4,869 critically ill adult patients with COVID-19 were enrolled into the trial between March 9, 2020, and June 22, 2021, from 197 sites in 14 countries. The final 180-day follow-up was completed on March 2, 2022.
As an adaptive platform trial, REMAP CAP continues to evaluate new interventions for the treatment of both COVID-19 and other non-COVID 19 respiratory tract infections.
REMAP-CAP is an Australian-led global adaptive trial investigating multiple treatments for COVID-19 among patients in Intensive Care Units (ICUs). Run by Monash University's Australian and New Zealand Intensive Care Research Centre, the trial mobilized to evaluate specific treatments for COVID-19 patients in ICUs in early March 2020.
More information: Long-term (180-Day) Outcomes in Critically Ill Patients With COVID-19 in the REMAP-CAP Randomized Clinical Trial, JAMA (2022). DOI: 10.1001/jama.2022.23257