Using spatial single-cell transcriptomics to visualize diffuse midline glioma
Children diagnosed with diffuse midline gliomas often die within a year after their initial diagnosis since there are no effective treatments yet for this rare cancer. But researchers now have a better understanding of what these tumors look like and can work towards developing new treatment approaches.
A team led by Mariella Filbin, MD, Ph.D., a physician at Dana-Farber/Boston Children's Cancer and Blood Disorders Center, has used spatial single-cell transcriptomics to visualize the structure of the tumor's cancer cells across different age groups and locations.
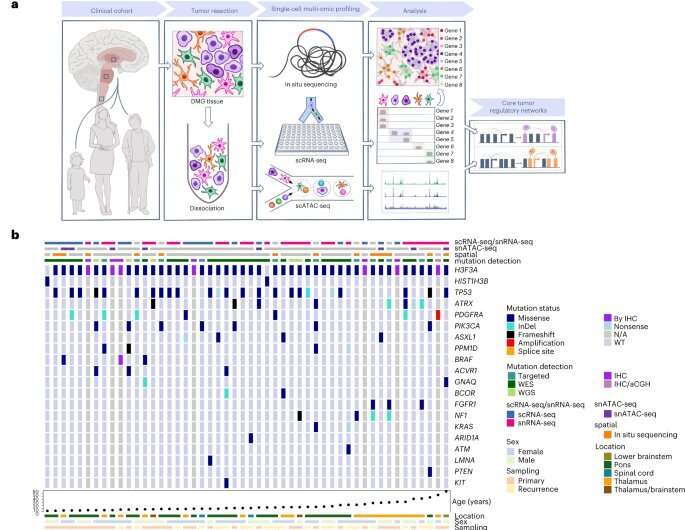
The researchers discovered that the spatial organization of cells could help explain why diffuse midline gliomas are so difficult to treat. The study, published in Nature Genetics on December 5, 2022, is the first to use spatially resolved single cell technology on patient tissues and highlights the need for future therapies to be specialized depending on the tumor's context.
The team focused their study on diffuse midline gliomas driven by a particular genetic change called a Histone3-K27M mutation that drives early stem cells of the brain to become cancerous.
Dr. Filbin and colleagues looked for age and location-dependent differences in the tumors by measuring all the genetic activity in tissue samples from 50 people, ranging from 2-68 years old, with the Histone3-K27M mutation.
The team found that pediatric tumors and tumors in the brainstem and spinal cord have more immature cells. Immature cells can proliferate quickly and easily because of their similarity to stem cells, which might help explain the lethality of diffuse midline gliomas in kids.
The researchers also noticed that adult tumors have more mesenchymal-like cells, commonly helpful for healing damaged tissues but can help aid tumor growth and progression in cancer. However, the researchers didn't detect any underlying genetic cause for this difference.
"Adult tumors seem to be shaped more by the immune microenvironment," explains Dr. Filbin. These findings suggest that kids and adults with diffuse midline gliomas might need different treatments.
Additionally, the researchers mapped where the genetic activity was in space to understand the tumor's structure and development. What they found was surprising: The cancer stem cells had formed "niches" by clumping together and surrounding themselves with a type of cell that can shield and protect the tumor.
"We now have a glimpse into how tumors visually organize themselves—walling off from the rest of the brain to prevent an immune attack and getting closer to cells that feed them," says Dr. Filbin. Understanding the structure of the tumor enables researchers to think about new treatment approaches.
Dr. Filbin now wants to understand how the different cancer cells talk to each other and form a barrier around the tumor so that her team can develop targeted treatments. "If we know how the cells communicate, perhaps we can block their signaling and prevent them from creating these niches," she says.
More information: Ilon Liu et al, The landscape of tumor cell states and spatial organization in H3-K27M mutant diffuse midline glioma across age and location, Nature Genetics (2022). DOI: 10.1038/s41588-022-01236-3