Tissue-specific immunity may be the future, if we can first learn its rules
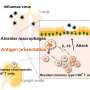
Recent pressure to maximize vaccine efficacy has stirred up many new discoveries within immunology, revealing numerous paradigms with untapped therapeutic potential. One growing branch of research is focused on tissue-resident memory T cells (TRM cells), a type of immune cell that provides long-lasting protection against pathogens attacking specific organs and tissues.
In a new study published December 28, 2022 in Immunity, scientists at University of California San Diego School of Medicine revealed a previously unappreciated complexity of TRM cell biology in the gut, which may inspire a new generation of precision therapeutics against infection, cancer and auto-immune disease.
After experiencing an infection, the immune system leaves behind memory T cells, which maintain a long-lasting molecular memory of the pathogen and are ready to sound the alarm if it ever returns. While some memory T cells are designed to circulate through the bloodstream and provide whole-body protection, others reside in specific organs and are specialized to fight the pathogens that target that part of the body. These TRM cells can provide life-long immunity at the target tissue, but can also contribute to autoimmune diseases if overactivated.
"TRM cells are the first responders, right at the front lines of infection," said senior author John T. Chang, MD, professor at UC San Diego School of Medicine. "Most of our vaccines are designed to provide systemic immunity, but we may be able to get even better protection by instead focusing on boosting the tissue-specific cells that encounter the pathogen first."
For example, a respiratory virus may be best fought by strengthening TRM cells in the nose and lungs, and a pathogenic gut microbe best treated by enhancing TRM cells in the intestines. Thus the goal is to develop therapeutics that could boost the formation and maintenance of TRM cells, or in the case of autoimmune disease, remove the immune cells by disrupting these same pathways.
The issue is, scientists still have a lot to learn about what helps TRM cells form and survive, and these rules may be quite different in each tissue type.
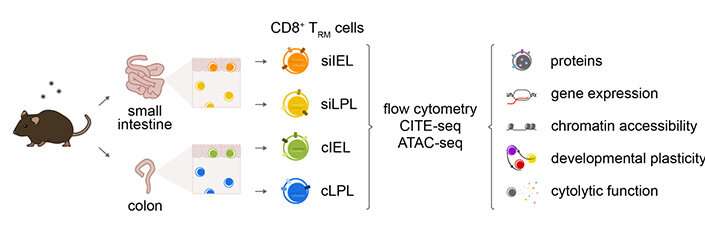
To explore this, the researchers performed a series of experiments to characterize TRM cells in mice from four different compartments of the gut: two organs (the small intestine and the colon) and two different tissue layers in each (the intraepithelial and lamina propria layers).
The experiments revealed that TRM cells in each tissue type exhibited distinct patterns of cytokine and granzyme expression, along with substantial transcriptional, epigenetic and functional heterogeneity. In other words, the same type of immune cells in each part of the gut appeared to be very different in their molecular makeup, function and the chemical signals they depend on.
Reinforcing this further, each population of cells also showed differential dependence on Eomesodermin (Eomes), a transcriptional factor known to affect TRM cell development. Eomes was canonically thought to repress TRM cells based on previous data collected from the skin, liver and kidney, but the new experiments revealed the opposite was true in the small intestine. There, Eomes proved to be surprisingly important in the survival of TRM cells. However, this was not the case in the colon, highlighting the high context-specificity even within the gut.
Future research will continue to define the rules of TRM cell formation and maintenance in other tissues and explore what drives their specificity. For example, the authors suggest that differences in the microbiome of the small intestine and the colon may contribute to the unique needs of their TRM cells, so manipulating the microbiome may be another approach to regulating immune cells in the gut.
"In the future, we want to be thinking about vaccines and other therapeutics that are tailored to the specific needs of each organ," said Chang. "By knowing what each tissue type needs to support the formation and maintenance of TRM cells, we can provide the most efficient immune defenses against disease."
More information: Yun Hsuan Lin et al, Small intestine and colon tissue-resident memory CD8+ T cells exhibit molecular heterogeneity and differential dependence on Eomes, Immunity (2022). DOI: 10.1016/j.immuni.2022.12.007