This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
From grave to cradle: Collagen-induced gut cell reprogramming
Most cells have a pretty normal life: they're born, they grow, they get old, and they die. But the Benjamin Buttons of the cellular world can go from old to young again in the right context. Now, researchers from Japan have identified a physical cue that sparks this change in cells in the human gut.
In a study published this month in Inflammation and Regeneration, researchers from Tokyo Medical and Dental University (TMDU) have revealed that the accumulation of a thick, extracellular material called collagen at injured sites in the gut stimulates cellular reprogramming.
When the intestine is injured, an inflammatory response occurs that is often associated with regeneration of the injured tissues. This process involves converting some mature intestinal cells back into fetal-like cells that can then generate new healthy tissue to repair the injured area.
"We previously showed that deposition of collagen at the site of intestinal injury promotes the conversion of intestinal/colonic epithelial cells covering the wound bed towards fetal-like progenitors in mice," says lead author of the study, Sakurako Kobayashi. "However, the detailed mechanism by which this occurs, and whether this process also occurs in humans, remained unclear."
To explore these questions, the researchers created collagen spheres, which are tiny balls of epithelial cells grown in purified collagen, from mouse and human intestinal cells. They then assessed gene expression in these spheres to clarify the mechanisms of inflammation-associated reprogramming.
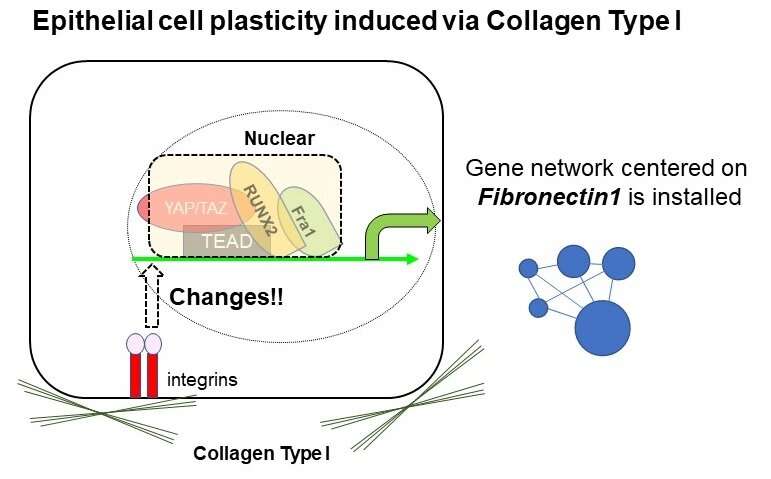
"The results showed that culturing in collagen induced the expression of inflammation-associated and fetal-like genes in both human and mouse intestinal cells," explains Shiro Yui, senior author. "As previously reported, the YAP/TAZ-TEAD axis definitely plays a central role in the induction of this distinctive gene expression signature, but this time we identified the cooperative transcriptional activity of Fra1 and RUNX2 in the process, which hammers the gene network centered on Fibronectin."
Importantly, the representative genes that were activated in the human collagen spheres were also highly expressed in tissue samples taken from inflamed regions of the gut in patients with ulcerative colitis.
"Taken together, our findings demonstrate that collagen has a significant influence on inflammation and cellular reprogramming in both mice and humans," says Kobayashi.
Given that some of the genes that were upregulated in the collagen spheres are also overexpressed in conditions such as colorectal cancer, it is possible that there is a link between the regenerative cascade and colorectal carcinogenesis. Thus, investigating the mechanisms of cell fate conversion using this model may enhance our understanding not only of how inflammation is influenced by the extracellular environment, but also how other disease processes occur in the gut.
More information: Sakurako Kobayashi et al, Collagen type I-mediated mechanotransduction controls epithelial cell fate conversion during intestinal inflammation, Inflammation and Regeneration (2022). DOI: 10.1186/s41232-022-00237-3