This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Simple spray could keep COVID away
What if preventing respiratory illnesses such as COVID-19 and influenza could be as easy as a quick spritz of nasal spray every morning?
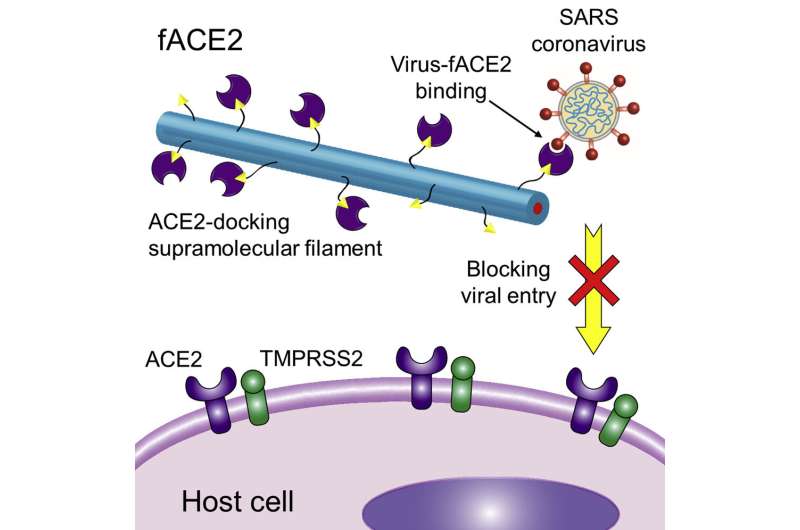
Numerous bacteria and viruses, including those that cause COVID-19 and influenza, enter the body through the lungs when people breathe, resulting in illness. Johns Hopkins engineers have created thin, thread-like strands of molecules called supramolecular filaments that are designed to be sprayed into the nose, blocking those harmful viruses from entering the lungs.
"The idea is that the filaments will work like a sponge to absorb the COVID-19 virus and other viruses before they have the chance to bind to cells in our airways. Even if the therapeutic can block the virus for an hour or two, that can be helpful when people must be in a public setting," said research team leader Honggang Cui, core researcher at the Institute for NanoBioTechnology and associate professor of chemical and biomolecular engineering at Johns Hopkins Whiting School of Engineering.
The team's results appeared recently in Matter, and the work was done through a collaboration with Hongpeng Jia, assistant professor of surgery, and other researchers at Johns Hopkins School of Medicine.
The key to this approach is the way that the filaments carry a receptor called angiotensin converting enzyme-2, or ACE2. These receptors are also found in cells in the nasal lining, the lung surface, and small intestine, and have many biological roles, such as regulating blood pressure and inflammation. The novel coronavirus enters our bodies primarily through interactions with this receptor. The virus's characteristic spike protein clicks into this receptor, much like a key going into a lock, allowing it to enter the cell and replicate. Once the virus is locked into the cell, it prevents the cell from executing its normal functions, leading to and exacerbating infections.
Researchers have long known that adding extra ACE2 into airways can block virus entry, essentially preventing the virus from binding with ACE2 in the lungs. However, since ACE2 has biological functions, simply delivering more ACE2 to the body may have unforeseeable complications. The research team's newly engineered filament, called fACE2, serves as a decoy binding site for the virus, with each filament offering several receptors for the COVID-19 spike protein to attach to, and silences ACE2's biological functions to avoid potential side effects.
"Our plan is that this would be administered as a nasal or oral spray, allowing it to be suspended in the lungs or settle on the surface of airways and lungs. When a person breathes in the COVID-19 virus, the virus will be fooled into binding to the decoy receptor and not the ACE2 receptors on cells," Cui said.
And because the filaments attract SARS-CoV-2's characteristic spike protein, it should work equally well on any current or future variants, the researchers predict.
The team tested its design in mouse models and found their filament was not only present in the rodents' lungs up to 24 hours later, but also elicited no inflammation or obvious damage to the lungs' structures, suggesting that fACE2 may be retained in the lungs for a period of time, and is safe.
While the team's original approach was to design a preventative therapeutic, they say that it also may have the potential to treat people with active COVID-19 infections by thwarting replication of newly acquired viruses.
"We think that fACE2 could also be used on other respiratory viruses that use the ACE2 receptor to infiltrate cells. The filament design is versatile and can be modified to carry various therapeutic proteins that target different receptors," Jia said.
More information: Caleb F. Anderson et al, Supramolecular filaments for concurrent ACE2 docking and enzymatic activity silencing enable coronavirus capture and infection prevention, Matter (2022). DOI: 10.1016/j.matt.2022.11.027