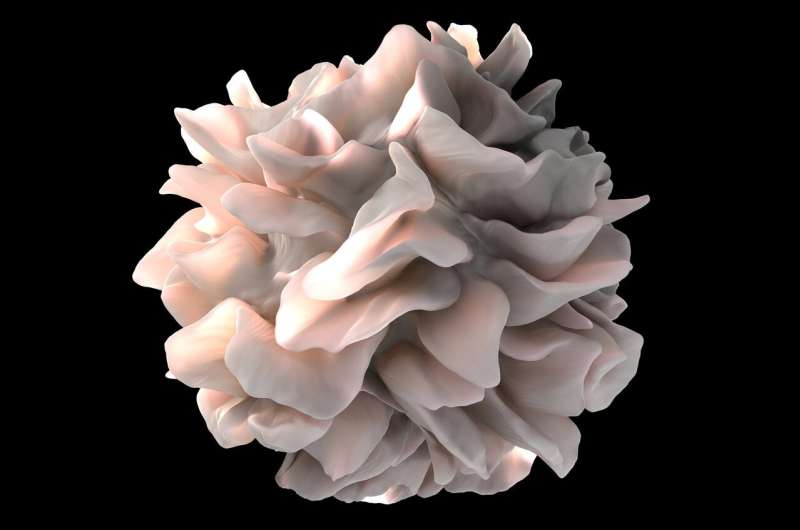
Artistic rendering of the surface of a human dendritic cell illustrating sheet-like processes that fold back onto the membrane surface. Credit: National Institutes of Health (NIH)
Immune checkpoint inhibitors that activate T cells to target cancer cells have greatly improved patient outcomes across a variety of tumor types. But not all patients respond to the therapies. Moffitt Cancer Center researchers are working to identify new treatment options for this subset of patients.
In a new study published in the journal Cancer Immunology Research, researchers, led by Amer A. Beg, Ph.D., show how stimulating dendritic cells through the CD40 and interferon β (IFNβ) pathways produces strong T cell activity against tumors and works in conjunction with immune checkpoint inhibitors to produce even stronger responses. The article also shares promising early results from a phase 1 clinical study of an oncolytic virus (MEM-288) that activates these pathways in patients with non-small cell lung cancer.
Dendritic cells play an important role in activating T cells to recognize infectious or foreign agents, including cancer cells. These characteristics suggest that dendritic cells may be an important cellular component in achieving optimum responses to checkpoint inhibitor therapy.
"Immune checkpoint inhibitors that target T cells have revolutionized cancer treatment, but there is increasing realization that targeting cell types responsible for T cell activation, such as dendritic cells, may be required for optimal patient benefit. However, the best strategy to target dendritic cells to unleash their full therapeutic potential has remained undefined," said Andreas N. Saltos, M.D., clinical research medical director in the Department of Thoracic Oncology at Moffitt and principal investigator of the MEM-288 oncolytic virus clinical trial.
Beg and a team of researchers focused their investigation on two separate pathways that are known to contribute to activation of dendritic cells, the CD40 and the IFNβ pathways. They performed laboratory experiments with an adenovirus that expressed the proteins IFNβ and a membrane-stable version of the CD40 ligand (MEM40).
They found that the combined expression of both MEM40 and IFNβ resulted in dendritic cell activation, maturation and migration to lymph nodes. Both MEM40 and IFNβ produced anti-tumor T-cell responses and reduced tumor growth in mouse models of melanoma and lung cancer, with stronger responses observed with combined expression of both proteins.
The researchers also demonstrated that direct tumor injection of the adenoviruses expressing MEM40 and IFNβ were able to reduce tumor growth within the injected tumor and reduce distant metastases, suggesting that dendritic cell activation by MEM40 and IFNβ can produce systemic anti-tumor effects. This combination virus treatment was effective in mouse cancer models that were insensitive to checkpoint inhibitors and could work in conjunction with checkpoint inhibitors to produce even stronger anti-tumor activity.
"We believe our findings point to a new therapeutic strategy to maximize dendritic cell-induced T-cell activation with potential to benefit patients as a single agent and in combination with immune checkpoint inhibitors," said Beg, senior member of the Department of Immunology at Moffitt.
Given these positive results, the Moffitt team worked in collaboration with Memgen, Inc., to create the MEM-288 oncolytic virus that drives expression of MEM40 and IFNβ. Oncolytic viruses preferentially replicate within cancer cells and lyse them, resulting in the release of antigens that promote further anti-tumor immunity. After testing their oncolytic virus in laboratory experiments, they initiated a phase 1 trial in patients with solid tumors to assess safety and efficacy.
Early results from non-small cell lung patients reveal that MEM-288 caused tumors to shrink in size and increased T cell numbers throughout the tumor environment, and importantly, the potential ability to generate systemic anti-tumor T cell immunity. The clinical trial is ongoing and continuing to recruit patients.
"The next phase of this study will be an expanded Phase 1b trial to investigate MEM-288 in combination with an immune checkpoint inhibitor in non-small cell lung cancer patients," said Mark Cantwell, Ph.D., chief scientific officer of Memgen and an author on this study. "We are excited to see early clinical activity, safety and generation of antitumor immunity by MEM-288, which may be crucial for synergy with immune checkpoint inhibitors."
The Phase 1b trial is expected to start in mid-2023.
More information: Hong Zheng et al, Combination IFNβ and membrane-stable CD40L maximize tumor dendritic cell activation and lymph node trafficking to elicit systemic T-cell immunity, Cancer Immunology Research (2023). DOI: 10.1158/2326-6066.CIR-22-0927