This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Real-world studies confirm effectiveness of bulevirtide to treat chronic hepatitis D
In 2020, bulevirtide (BLV) was conditionally approved for treating chronic hepatitis delta (CHD), an inflammation of the liver caused by hepatitis D virus (HDV). Now real-world studies of patients treated outside of clinical trials confirm that long-term suppressive therapy with BLV monotherapy has the potential to reduce viral replication and improve liver tests of these difficult-to-treat patients for the first time in 45 years, report investigators in the Journal of Hepatology and its companion journal JHEP Reports.
Two of the studies, led by Pietro Lampertico, MD, Ph.D., Division of Gastroenterology and Hepatology, Foundation IRCCS Ca' Granda Ospedale Maggiore Policlinico, Milan, Italy, were designed to assess the effectiveness and safety of patients with advanced HDV-related compensated cirrhosis being treated with BLV 2mg monotherapy and the consequences of discontinuing this treatment.
"HDV is the most severe form of chronic viral hepatitis," explained Dr. Lampertico. "For many years, the only therapeutic option was the off-label administration of pegylated-interferon-alpha (PegIFNa), an approach characterized by suboptimal efficacy, an unfavorable safety profile and several contraindications."
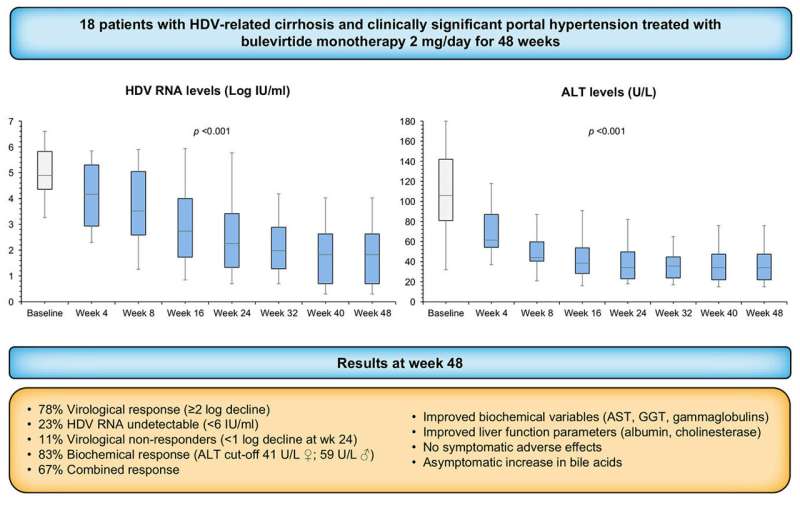
In a study of 18 patients with HDV-related advanced cirrhosis treated with BLV 2 mg/day for 48 weeks, Dr. Lampertico and colleagues demonstrated significant virological, biochemical and combined response rates associated with improvement of liver function.
"The efficacy and safety of BLV monotherapy in patients with advanced compensated cirrhosis were unknown before this study. Virological and biochemical responses to BLV monotherapy that we observed in our difficult-to-treat patients with HDV-related compensated cirrhosis were similar to those shown in the phase III registration study," Dr. Lampertico noted.
In a case report, Dr. Lampertico and co-investigators demonstrated that HDV could be successfully eradicated from both serum and liver following a three-year course of BLV monotherapy, despite the persistence of HBsAg, in a patient with HDV-related compensated cirrhosis and esophageal varices. During the 72-week off-BLV follow-up, liver biopsy, intrahepatic HDV RNA and hepatitis D antigen were undetectable, less than 1% of hepatocytes were HBsAg positive and all were negative for hepatitis B core antigen.
"We were surprised to demonstrate that HDV can be eradicated following a finite course of an entry inhibitor administered as monotherapy such as BLV 2mg/day, despite the persistence of HBsAg positivity," commented Dr. Lampertico.
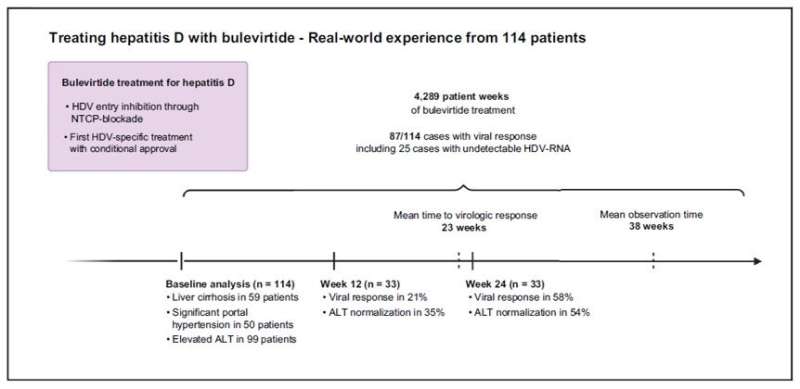
In a study in JHEP Reports led by PD Dr. med. Katja Deterding, MD, Department. of Gastroenterology, Hepatology and Endocrinology at Hannover Medical School, Hannover, Germany, investigators report the first data from the largest multicenter cohort of patients to date who were treated with BLV under real-world conditions, including 50 patients with signs of significant portal hypertension, elevated pressure in the major vein that leads to the liver.
The retrospective analysis of 114 cases covered 4,289 patient weeks of BLV treatment. Viral response was observed in 87 cases while hepatic inflammation improved, and treatment was well tolerated. More than 50% of patients showed a virologic response with less than 10% of patients not achieving an HDV RNA drop of at least 90% after 24 weeks. An improvement of biochemical hepatitis activity as measured by the liver enzyme alanine transaminase (ALT) values was observed regardless of virologic response. Investigators concluded that treatment was safe and well tolerated and associated with improvements in liver cirrhosis and portal hypertension with prolonged treatment.
"In line with other real-world cohorts and clinical trials our real-world study confirms the antiviral activity of BLV," noted Dr. Deterding. "We were surprised to see an improvement in biochemical hepatitis activity even in cases without viral response. Potential explanations for this phenomenon include anti-inflammatory properties of BLV."
"This is the first time that patients with HDV-related chronic advanced liver disease can be treated with an antiviral therapy since 1977 when HDV was discovered. Long-term suppressive therapy with BLV 2 mg/day has the potential to improve survival, of these difficult-to-treat patients for the first time in 45 years," concluded Dr. Lampertico. "We also found that BLV treatment can be successfully discontinued in some HDV patients who achieved long-term viral suppression while on therapy."
HDV infection occurs when people become infected with both hepatitis B and D virus either simultaneously (co-infection) or acquire the hepatitis D virus after first being infected with hepatitis B (super-infection). According to the World Health Organization, HDV affects nearly 5% of individuals with a chronic infection resulting from hepatitis B virus (HBV). Populations that are more likely to have HBV and HDV co-infection include Indigenous populations, recipients of hemodialysis and individuals who inject drugs.
More information: Elisabetta Degasperi et al, Bulevirtide monotherapy for 48 weeks in patients with HDV-related compensated cirrhosis and clinically significant portal hypertension, Journal of Hepatology (2022). DOI: 10.1016/j.jhep.2022.07.016
Maria Paola Anolli et al, A 3-year course of bulevirtide monotherapy may cure HDV infection in patients with cirrhosis, Journal of Hepatology (2023). DOI: 10.1016/j.jhep.2022.12.023
Christopher Dietz-Fricke et al, Treating hepatitis D with bulevirtide—Real-world experience from 114 patients, JHEP Reports (2023). DOI: 10.1016/j.jhepr.2023.100686