This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Mutation in ribosomal protein in heart and skeletal muscle leads to impaired cardiac contractility in mice
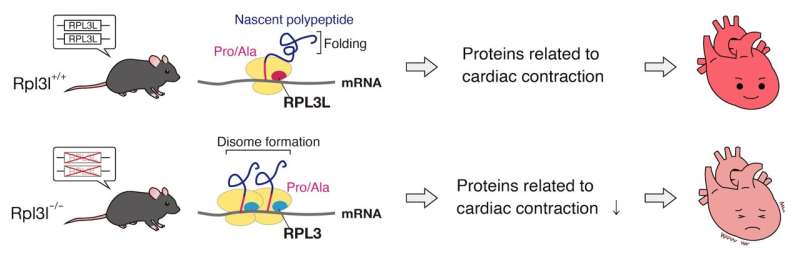
A team of researchers have discovered that a mutation in a ribosomal protein found specifically in heart and skeletal muscle leads to impaired cardiac contractility in mice.
The mutation was found to delay the rate of translating mRNA, leading to ribosomes colliding and causing protein folding abnormalities. The abnormal proteins would then be targeted and degraded by the cell's quality control system. Moreover, while the deficiency in the ribosomal protein, known as RPL3L, altered translation dynamics for the entire tissue, its effects were most pronounced for proteins related cardiac muscle contraction.
The study, published in Nature Communications, shines new light into the dynamics of a molecule as fundamental as a ribosome. Furthermore, since RPL3L gene deficiencies have been found in humans with cardiomyopathy and atrial fibrillation, the team hopes their new findings can lead to future treatments.
For cells to produce the proteins and molecules that make the body function, DNA is transcribed into messenger RNA, or mRNA, which is then used as a blueprint to link amino acids together and build a protein. At the heart of the protein building process is the ribosome, which reads the mRNA and translates that code into proteins.
Because of its fundamental function, ribosomes are found within all cells, and they were thought to be generally the same. However, recent studies have revealed the existence of differences in ribosomal structures.
"These differences in a ribosomal structure have shown to lead to translation specificity. For example, some ribosomes are better at producing proteins that control metabolism, or the cell cycle. It's a new concept called Ribosome Heterogeneity," explains Keiichi I. Nakayama of Kyushu University's Medical Institute of Bioregulation who led the study. "We hypothesized that this heterogeneity exists between tissues. After screening for tissue-specific ribosomal proteins we found one that was only expressed in heart and skeletal muscle: RPL3L."
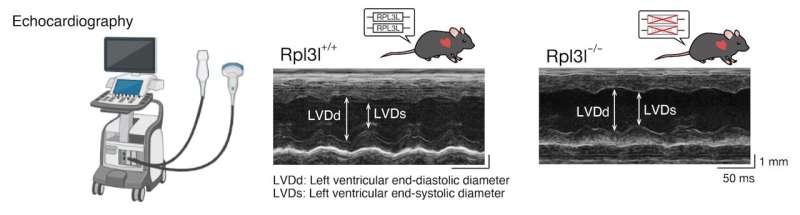
To elucidate the function of RPL3L, the team studied the hearts of mice with a mutated RPL3L gene. As expected, echocardiographic analysis showed that they had reduced cardiac contractility. Their next step was to study why exactly this mutation led to such a condition. As it turns out, the RPL3L mutation was causing a "translational traffic jam" for proteins critical in proper heart function.
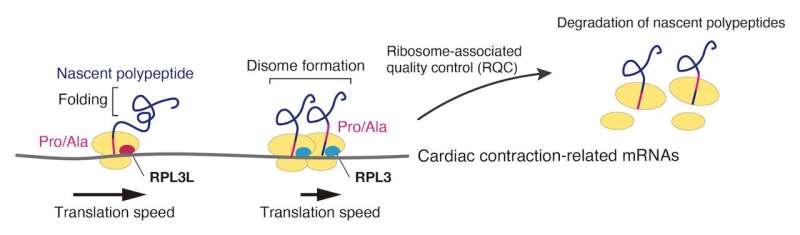
"We found that the mutant RPL3L would delay translation for the proline and alanine codons on mRNA. This delay caused ribosomes to collide, resulting in proteins not folding correctly," continues Nakayama. "Misfolded proteins would then be cleared out from the cell by its quality control system. More importantly, many of the misfolded proteins were ones involved in cardiac contraction."
The team hopes that by deepening our understanding of the translation dynamics of ribosome such as RPL3L, they can better understand how its genetic mutations—found in patients with dilated cardiomyopathy and atrial fibrillation—can lead to heart disease.
"We are developing new understandings in the field of biology and medicine every day, even in something as fundamental as ribosomes. I'm exciting to see what we'll find next," concludes Nakayama.
More information: Chisa Shiraishi et al, RPL3L-containing ribosomes determine translation elongation dynamics required for cardiac function, Nature Communications (2023). DOI: 10.1038/s41467-023-37838-6