This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New neurodevelopmental disorder identified among patients with common symptoms
A new type of developmental disability caused by mutations in a gene known as CBX1 has been discovered by a UT Southwestern Medical Center researcher and his colleagues. The findings, reported in Genetics in Medicine, offer insight into the role this gene plays in development and could eventually lead to therapies for a range of related disorders.
"Genetic testing approaches are able to make a diagnosis in only about a third of patients with developmental disabilities. That leaves another two-thirds with no explanation for their condition. This study is a success story in which we linked a gene never associated with human disease until now with a neurodevelopmental disorder," said Kosuke Izumi, M.D., Ph.D., Assistant Professor of Pediatrics at UT Southwestern, who led the study while previously serving at Children's Hospital of Philadelphia. Dr. Izumi is a new member of the Division of Pediatric Genetics and Metabolism in the Department of Pediatrics at UT Southwestern.
In 2017, Dr. Izumi and his colleagues met a patient with a unique set of symptoms: developmental delay, low muscle tone, features of autism spectrum disorder, an unusually broad forehead, and an above-average head circumference. Genetic testing revealed that this patient had mutations in CBX1, a gene that produces a protein called HP1β.
Previous studies showed that this protein mediates the interactions of other proteins within heterochromatin, a condensed form of DNA seen in the cellular nucleus. Although some of the proteins that HP1β interacts with have been associated with developmental disorders, Dr. Izumi explained, HP1β itself has never been connected with any health conditions.
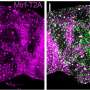
Dr. Izumi was able to find two other research teams that had encountered patients with different mutations in CBX1 and who shared the same unique symptoms. Molecular modeling experiments showed that the newly identified CBX1 mutations affected HP1β's ability to bind to chromatin. The findings were supported by experiments in cells derived from the patients. When the researchers generated mice that also carried these mutations, they found that HP1β distributed abnormally throughout cell nuclei. These mutant animals also displayed aberrant behaviors, such as a lag in the time it took them to respond to a startling noise.
Although the three patients in this study are the only ones identified so far with this neurodevelopmental disability, Dr. Izumi said researchers have found several other diseases linked to the proteins that interact with HP1β. These diseases, which share features with the three patients, are relatively rare, he explained, but together they account for a significant number of patients worldwide.
"By combining genetic diagnoses that could be related, the incidence together is not that low," said Dr. Izumi, who has an appointment in the Children's Medical Center Research Institute at UT Southwestern. "I'm optimistic that once we identify the pathway that these diseases may share, we might be able to target them with a common treatment."
Dr. Izumi performed the majority of the work for this study as a principal investigator at Children's Hospital of Philadelphia.
More information: Yukiko Kuroda et al, Dominant-negative variants in CBX1 cause a neurodevelopmental disorder, Genetics in Medicine (2023). DOI: 10.1016/j.gim.2023.100861