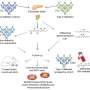
A research team from the Cedars-Sinai Heart Institute, Johns Hopkins University and China Medical University and Hospital in Taiwan have described for the first time the mechanisms by which variants of a specific gene, CAPON or NOS1AP, can disrupt normal heart rhythm. Until recently, CAPON was not even suspected of existing in heart tissue or playing a role in heart function.
The study, conducted in guinea pigs, confirms that CAPON naturally exists in the ventricles (pumping chambers) of the heart. The researchers show that CAPON interacts with a signaling molecule (NOS1) in heart muscle to influence signaling pathways and modify cell-to-cell interactions (calcium ion and potassium ion channels) that control electrical currents.
Eduardo Marbán, M.D., Ph.D., director of the Cedars-Sinai Heart Institute, is senior author of an article, published online March 4 in Proceedings of the National Academy of Sciences (Early Edition), that fully describes these events.
The effects of CAPON and its variants are seen in the “QT interval” of EKGs, which reflect electrical activity from the time the ventricles are stimulated to the end of the heart muscle activity in a single heartbeat. Whether too long or too short, QT interval abnormalities can represent serious heart rhythm problems, including the risk of sudden death.
Long- and short-QT syndromes can be caused by rare congenital disorders that affect the ion channels, but most deaths caused by sudden arrhythmias occur in people who do not have these genetic mutations. Until recently, physicians and researchers were unable to explain the basis of QT interval abnormalities in otherwise healthy people.
In 2006, however, Marbán was among researchers who used a new approach to gene discovery to search for genetic influences on QT interval variations. From a general population which has been extensively studied (the KORA Cohort in Germany), the researchers identified subjects who had long- or short-QT intervals. Studying the genetic makeup of those individuals, they discovered an association between QT intervals and the CAPON gene. Their findings, published in Nature Genetics, were surprising because CAPON, while known for its involvement in brain nerve cells, was not expected to exist in heart tissue.
Subsequent studies, including this one, have confirmed the existence of CAPON in heart tissue and illuminated its effects on heart function. According to the PNAS article, the new “findings provide a rationale for the association of CAPON gene variants with extremes of the QT interval in human populations.”
Source: Cedars-Sinai Medical Center