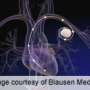
(HealthDay)—An implanted wireless device that measures key vital signs in people with heart failure has been approved by the U.S. Food and Drug Administration.
The CardioMEMSHF system measures pulmonary artery pressure and heart rates and sends this information remotely to the patient's doctors, the FDA said Wednesday in a news release.
The device is intended for people with New York Heart Association Class III heart failure, the FDA said. Some 5.8 million people in the United States have heart failure, in which the heart can't pump enough blood. People with NYHA Class III heart failure have difficulty performing everyday tasks such as walking short distances, the agency said.
The device was evaluated in a clinical study involving 550 people. All devices implanted were still working after six months, the FDA said.
The agency said it is requiring a post-approval study to gather information about the device's long-term performance.
The system is manufactured by CardioMEMS Inc., based in Atlanta.
More information: Visit the FDA to learn more about this approval.
Copyright © 2014 HealthDay. All rights reserved.