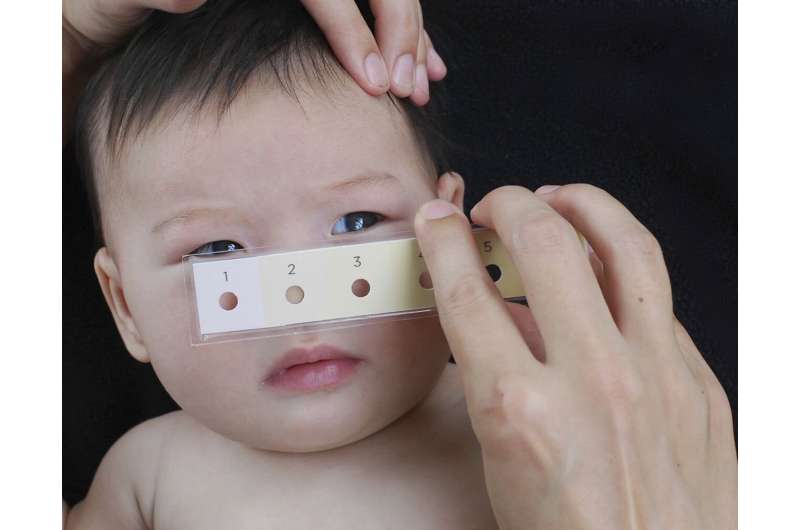
Closeup view of the bili-ruler being used on an infant. Credit: Brigham and Women's Hospital
Neonatal hyperbilirubinemia, commonly known as newborn jaundice, is a condition that affects up to 80 percent of newborns in the first week of life. Severe hyperbilirubinemia (bilirubin levels >20mg/dL) affects an estimated 1 million infants around the world annually. If diagnosed early, severe hyperbilirubinemia is largely treatable with high-intensity phototherapy, resulting in recovery without long-term consequences. If left undiagnosed or untreated, severe hyperbilirubinemia may lead to irreversible brain damage or even death. These complications often arise in low-resource settings; in low- and middle-income countries (LMICs), one-third of infants with extreme hyperbilirubinemia die from a condition that is largely treatable.
But access to treatment itself may not be the primary challenge. One significant barrier to managing hyperbilirubinemia in LMICs is the failure to recognize the severity of the condition before the onset of irreversible symptoms. A team from Brigham and Women's Hospital recently reported the creation and validation of a novel tool, the Bili-ruler, designed for use by frontline health workers to screen for hyperbilirubinemia in low-resource settings. The findings are published in Pediatrics.
"In low-income settings, many people don't have access to very basic diagnostics, medications and interventions that could prevent a large burden of neonatal morbidity and mortality," said lead author Anne CC Lee, MD, MPH, a pediatrician, director of the Brigham Global Newborn Health Lab and principal investigator of the project. "Improving early recognition and care seeking for potentially serious newborn illness is a first step."
The first low-cost screening tool for jaundice was the Gosset icterometer developed in the 1950s. This device comprised five shades of yellow and presented good correlation with serum bilirubin concentrations; however, it was developed using robbialac paint which led to a lack of color standardization and was never widely adopted. Currently, in high-income settings, there are trancutenous bilirubin instruments, but these cost over $7,000 and are not affordable in most low-income settings.
"Ultimately, if the baby is not diagnosed or is diagnosed too late, there is very little that can be done before the effects are permanent," said co-author Lian Folger, a program coordinator at the Global Newborn Health Lab.
To address this barrier of illness recognition, the team designed the Bili-ruler to improve the identification of clinically severe hyperbilirubinemia in low-resource settings. As its name suggests, the Bili-ruler is a portable, handheld "ruler" to measure the degree of newborn jaundice, using six color strips arranged in a stepwise gradient of increasing yellow hue. The colors were developed using advanced digital color processing of images of infants with different levels of hyperbilirubinemia.
To assess for jaundice, the Bili-ruler is pressed against the infant's skin, and the underlying skin tone is visualized through a circular window enclosed by a uniform color strip. This process is repeated for all six colors, and the user chooses the score which corresponds to the color that most closely matches the underlying skin tone. The team went through many iterations of the design of the device and collaborated with groups at MIT that specialize in visual design.
To validate the Bili-ruler across diverse populations, the research team recruited a total of 790 newborns from the Brigham and its partner institution, the Sylhet Osmani Medical College Hospital (SOMCH) in Sylhet, Bangladesh. Newborns were eligible for inclusion if they were <28 days old, generally healthy, and had not previously received phototherapy or exchange transfusion.
In the study, the Bili-ruler was used to assess for jaundice alongside clinical measures of transcutaneous bilirubin (TcB) and total serum bilirubin (TSB) levels. Bili-ruler measurements were obtained on the forehead, nose, abdomen, palms, or soles of feet, without previous knowledge of bilirubin levels. The scores were then compared with TcB and TSB.
The Bili-ruler had high diagnostic accuracy and validity, showing strongest positive correlations between measurements taken on the nose with bilirubin concentrations measured by both TcB and TSB. In addition, measurements on the nose had high validity, sensitivity and specificity for identifying hyperbilirubinemia of several different thresholds. Bili-ruler scores on the foot also had high sensitivity, but lower specificity in identifying serum bilirubin at very high thresholds.
Results initially seemed consistent between infants of varying skin tones—an important consideration for use in multi-ethnic populations. However, since the largest populations validated in the study were south Asian and white, the researchers hope to expand validating the Bili-ruler in other ethnicities—namely Hispanic and black populations.
Currently, the team is working to partner with organizations in Ghana and Peru to expand the application and validity of the device. In Peru, the researchers are working with Little Sparrow Technologies, INMED, and Bilimetrix to implement a low-cost, package to guide both diagnostic evaluation and clinical management of jaundice.
Lee and Folger speak passionately about the significance and global health impact of their work.
"The hope is that this tool will improve the early recognition and more accurate diagnosis of severe jaundice in settings that traditionally do have not had access to care. Two of my own children had severe hyperbilirubinemia and required phototherapy. They likely would not have had the same healthy outcome if they were born at home and without postnatal care in rural Bangladesh. We hope that the Bili-ruler will improve access to treatment for hyperbilirubinemia and improve health outcomes for babies born in low-resource settings," said Lee. "Every baby, no matter where they are born, deserves the equal opportunity for a healthy start in life."
More information: Anne CC Lee et al, A Novel Icterometer for Hyperbilirubinemia Screening in Low-Resource Settings, Pediatrics (2019). DOI: 10.1542/peds.2018-2039
Journal information: Pediatrics
Provided by Brigham and Women's Hospital