A leading group of Alzheimer's researchers contends that, as biomarkers to detect signals of the disease improve at providing clinically meaningful information, researchers will need guidance on how to constructively disclose test results and track how disclosure impacts both patients and the data collected in research studies. A survey conducted by a group including experts from the Perelman School of Medicine at the University of Pennsylvania found that a majority of Alzheimer's researchers supported disclosure of results to study participants. The study is published online in Neurology.
"While this is not a call to immediately tell subjects their biomarker results, it does show that the field is moving to a point where experts want to share valid and meaningful results with participants," said co-senior author Jason Karlawish, MD, professor of Medicine and Medical Ethics and Health Policy. "As we gain more data on the predictive abilities of these measurements, we will need models and methods to effectively reveal results."
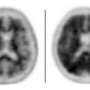
The study surveyed 139 Alzheimer's clinical trial leaders and coordinators from the Alzheimer's Disease Neuroimaging Initiative (ADNI) in April 2012, just before the U.S. Food and Drug Administration approved the amyloid-binding radiotracer known as Amyvid (florbetapir). 73 percent of respondents supported disclosing amyloid imaging results to study participants with mild cognitive impairment, whereas 58 percent supported giving amyloid imaging results to those with normal cognition.
Six themes emerged from the survey, regarding participant preferences and cognition levels, researchers' requests to develop standardized counseling procedures, participant education, and standardization of data-gathering, and concerns regarding potential harms and benefits to participants, as well as the ways disclosure could impact study results.
Currently, ADNI has a policy to not disclose results to participants, but the survey showed a growing trend of experts who would favor revising this policy. In addition to finding amyloid imaging results valuable, Alzheimer's experts also valued other biomarker data collected in ADNI, such as spinal fluid tests, PET imaging, and other psychometric tests, suggesting that if amyloid imaging results were allowed to be disclosed, it would likely lead to disclosure of other test results.
Journal information: Neurology
Provided by University of Pennsylvania School of Medicine