The compelling urge to satisfy one's hunger enlists structures throughout the brain, as might be expected in a process so necessary for survival. But until now, studies of those structures and of the feeding cycle have been only fragmentary--measuring brain regions only at specific times in the feeding cycle.
Now, however, Ivan de Araujo, Duke University Medical Center, and colleagues report they have mapped the activity of whole ensembles of neurons in multiple feeding-related brain areas across a full cycle of hunger-satiety-hunger. Their findings, reported in the August 17, 2006, issue of the journal Neuron, published by Cell Press, open the way to understanding how these ensembles of neurons integrate to form a sort of distributed "code" that governs the motivation that drives organisms to satisfy their hunger.
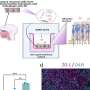
In their paper, Ivan de Araujo and colleagues implanted bundles of infinitesimal recording electrodes in areas of rat brain known to be involved in feeding, motivation, and behavior. Those areas include the lateral hypothalamus, orbitofrontal cortex, insular cortex, and amygdala. The researchers then recorded neuronal activity in those regions through a feeding cycle, in which the rats became hungry, fed on sugar water to satisfy that hunger, and then grew hungry again.
"This allowed us to measure both the ability of single neurons to encode for specific phases of a feeding cycle and how neuronal populations integrate information conveyed by these phase-specific neurons in order to reflect the animal's motivational state," wrote the researchers.
By isolating and comparing signals from particular neurons in the various regions at various times in the cycle, the researchers gained insight into the roles neurons in those regions played in feeding motivation and satisfaction, they wrote. The researchers found that they could, indeed, distinguish neurons that were sensitive to changes in satiety states as the animals satisfied their hunger. They could also measure how populations of neurons changed their activity over the different phases of a feeding cycle, reflecting the physiological state of the animals.
Importantly, they found that measuring the activity of populations of neurons was a much more effective way of measuring the satiety state of an animal than measuring activity of only individual neurons in an area. And the more neurons they included in such populations, the more accurate the measure of that satiety state, they found.
Araujo and colleagues concluded that their analysis showed that while single neurons were preferentially responsive to particular phases in the metabolic status of the animal as it went through a hunger-satiety-hunger cycle, "when combined as ensembles, however, these neurons gained the ability to provide a population code that allows for predictions on the current behavioral state (hunger/satiety) of the animal by integrating information conveyed by its constituent units."
"Our results support the hypothesis that while single neurons are preferentially responsive to variations in metabolic status, neural ensembles appear to integrate the information provided by these neural sensors to maintain similar levels of activity across comparable behavioral states," they concluded. "This distributed code acting across separate hunger phases might constitute a neural mechanism underlying meal initiation under different peripheral and metabolic environments," they wrote.
Source: Cell Press