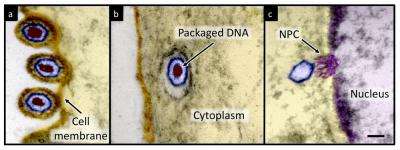
This is an electron micrograph of herpes simplex virus type 1 infecting a cell. Carnegie Mellon researchers have for the first time calculated the pressure inside the virus that powers the ejection of viral DNA into a host cell. Credit: Carnegie Mellon University
Herpes viruses are like tiny powder kegs waiting to explode. For more than 20 years scientists suspected that herpes viruses were packaged so full of genetic material that they built up an internal pressure so strong it could shoot viral DNA into a host cell during infection. No one had been able to prove that theory until now.
Carnegie Mellon University biophysicist Alex Evilevitch together with his graduate student David Bauer and University of Pittsburgh collaborators Fred Homa and Jamie Huffman have measured, for the first time, the pressure inside human herpes simplex virus 1 (HSV-1). The study, published online in the Journal of the American Chemical Society, provides the first experimental evidence of high internal pressure within a virus that infects humans. This phenomenon was previously only attributed to viruses that infect bacteria, called bacteriophages.
"Despite billions of years of evolution separating eukaryotic viruses and bacteriophages, the existence of an internal pressure capable of powering the ejection of DNA into a host cell has been conserved. This suggests that it is a key mechanism for viral infection across organisms and presents us with a new drug target for antiviral therapies," said Evilevitch, associate professor of physics.
Current antiviral medications are highly specialized. They target molecules essential to the replication cycle of specific viruses, so each medication is designed to treat only one type of viral infection. Additionally, since viruses mutate over time, they can become less susceptible to targeted medications. But designing drugs aimed at stopping a fundamental mechanism of viral infection, in this case the tightly packed genome that causes such high internal pressure, could yield new classes of broad-based, mutation-resistant antiviral treatments, according to Evilevitch.
Many viruses, whether they infect bacteria, plants or animals, are adept at packing long stretches of nucleic acid (DNA or RNA) within their nanometer-sized protein shells. HSV-1 contains double-stranded DNA that is 400 times longer than the radius of the viral shell it fills. The DNA gets packaged so tightly that it bends upon itself, resulting in repulsive forces that exert tremendous energy and pressure on the virus's outer shell. When HSV-1 infects a cell, it enters through the outer cell membrane and makes its way to the cell nucleus, where it docks in a small hole found in the nuclear membrane. That act is like a key opening a lock that allows the HSV-1 to eject its DNA into the cell nucleus.
For years scientists hypothesized that a high internal pressure inside eukaryotic viruses like HSV-1 enabled the virus to shoot its DNA into the host cell's nucleus. But it was impossible to measure HSV-1's internal pressure without knowing how to release the viral DNA in a controlled, experimental setting. Evilevitch's colleagues William Newcomb and Jay Brown, professors of microbiology and cancer biology at the University of Virginia's School of Medicine, discovered protein called UL6 that, when degraded, caused DNA to exit the virus. In his experiment, Evilevitch and Bauer degraded UL6, which triggered HSV-1 to eject its DNA. At the same time, they applied an external force to the virus and monitored how much DNA was released. When the external pressure reached tens of atmospheres, HSV-1 didn't release any DNA, indicating that the internal and external pressure were equal.
"This measurement of high internal pressure - tens of atmospheres - within a human virus is a fundamentally important discovery of the physical-chemical mechanism of eukaryotic viral infection," Evilevitch said.
The mechanism of pressurized infection found in HSV-1 is applicable to any of the eight known Herpes viruses that cause disease in humans, including Varicella zoster virus, which causes chickenpox in children and shingles in adults, and the Epstein-Barr virus, which causes mononucleosis and is associated with several types of cancer, such as Hodgkin's lymphoma.
More information: pubs.acs.org/doi/abs/10.1021/ja404008r
Journal information: Journal of the American Chemical Society
Provided by Carnegie Mellon University