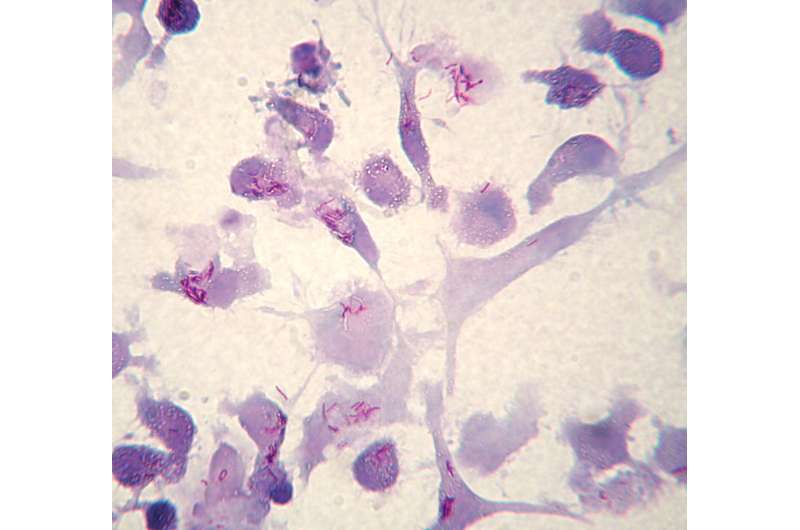
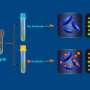
Mycobacterium tuberculosis (Mtb) infected Human Dendritic Cells. Mtb appear as isolated or grouped red-purple stained rods. Kinyoun staining. Optical mycrocoscopy 20x. Credit: Roberto Nisini
(Medical Xpress)—An international team of researchers has developed a urine test that can be used to detect tuberculosis (TB) in human patients. In their paper published in Science Translational Medicine, the group describes how they developed the test and how well it works.
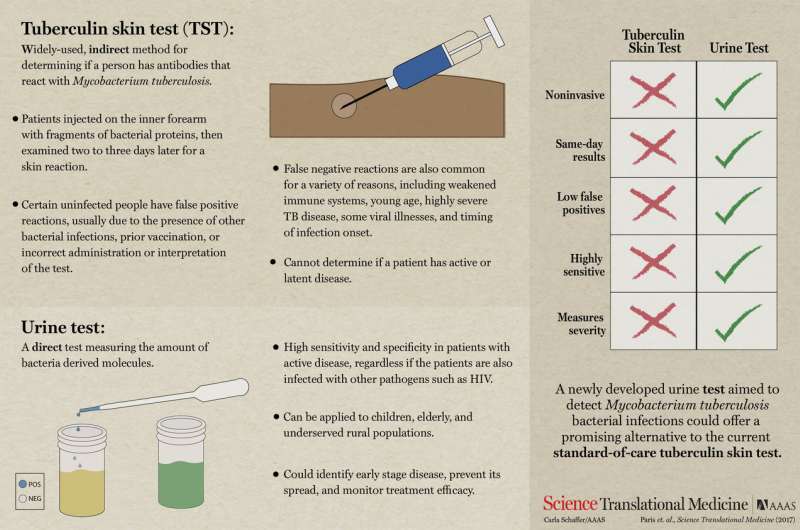
Scientists have been trying for some time to develop an easy way to test people for a Mycobacterium tuberculosis infection. The two main types of tests currently in use, the skin test and the culture test, require a high degree of expertise. In this new effort, the researchers claim to have developed a urine test that can be conducted by untrained health care workers.
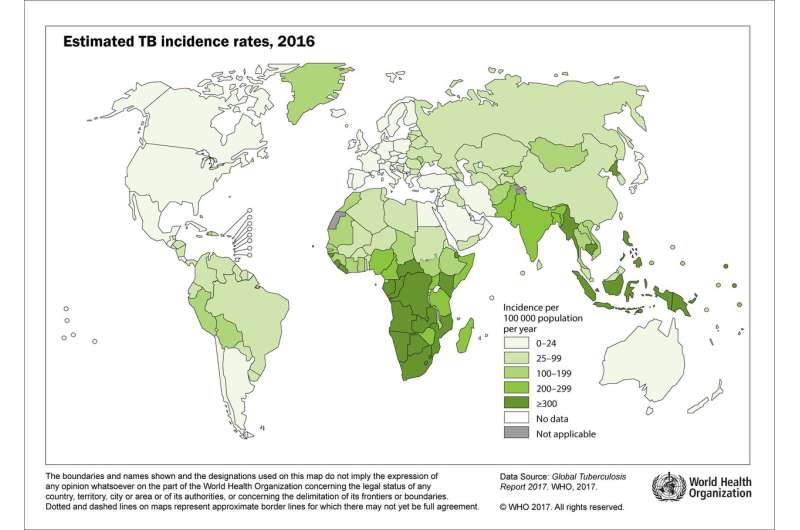
TB is a bacterial infection that primarily infects the lungs—10 million people are infected annually, and more than a 1.5 million of those victims die. Earlier detection and treatment, it is believed, would reduce suffering and fatalities. That is why scientists have been working so hard to find a method to detect it in people in a way that doesn't involve a lot of skill—such a test could be administered in third-world outposts, where health care is often lacking.
The new test works, the group explains, by employing tiny molecular cages that trap a type of sugar that coats the tuberculosis bacteria using a type of a dye.
The researchers report that the test detects TB at both low and high sugar levels, which means it can be used to test people at virtually any stage of the disease. They also report using the new test on 48 people in Peru known to have TB and found it has greater than 95 percent sensitivity. Furthermore, they report that the test works on people who are not HIV positive—all earlier urine tests for TB only worked for people who also had HIV infections, likely because their compromised immune systems allowed very high concentrations of the bacteria to grow. The team also reports that early indications have shown that the test can also offer an approximate degree of severity of infection—higher sugar levels mean more bacteria are active.
Infographic explaining the advantages of a new test for tuberculosis. Credit: Carla Schaffer / L. Paris et al. / AAAS
The team plans to continue improving their test, including devising a means for allowing field testing of people given treatments to see how well they respond—they also plan to keep looking to see if there are other markers they can trap that will give away the presence of the bacteria.
Map showing worldwide prevalence of tuberculosis infections. Credit: L. Paris et al., Science Translational Medicine (2017)
Interview clip of Alessandra Luchini, Associate Professor, George Mason University College of Science. Credit: George Mason University
More information: Luisa Paris et al. Urine lipoarabinomannan glycan in HIV-negative patients with pulmonary tuberculosis correlates with disease severity, Science Translational Medicine (2017). DOI: 10.1126/scitranslmed.aal2807
Abstract
An accurate urine test for pulmonary tuberculosis (TB), affecting 9.6 million patients worldwide, is critically needed for surveillance and treatment management. Past attempts failed to reliably detect the mycobacterial glycan antigen lipoarabinomannan (LAM), a marker of active TB, in HIV-negative, pulmonary TB–infected patients' urine (85% of 9.6 million patients). We apply a copper complex dye within a hydrogel nanocage that captures LAM with very high affinity, displacing interfering urine proteins. The technology was applied to study pretreatment urine from 48 Peruvian patients, all negative for HIV, with microbiologically confirmed active pulmonary TB. LAM was quantitatively measured in the urine with a sensitivity of >95% and a specificity of >80% (n = 101) in a concentration range of 14 to 2000 picograms per milliliter, as compared to non-TB, healthy and diseased, age-matched controls (evaluated by receiver operating characteristic analysis; area under the curve, 0.95; 95% confidence interval, 0.9005 to 0.9957). Urinary LAM was elevated in patients with a higher mycobacterial burden (n = 42), a higher proportion of weight loss (n = 37), or cough (n = 50). The technology can be configured in a variety of formats to detect a panel of previously undetectable very-low-abundance TB urinary analytes. Eight of nine patients who were smear-negative and culture-positive for TB tested positive for urinary LAM. This technology has broad implications for pulmonary TB screening, transmission control, and treatment management for HIV-negative patients.
Journal information: Science Translational Medicine
© 2017 Medical Xpress