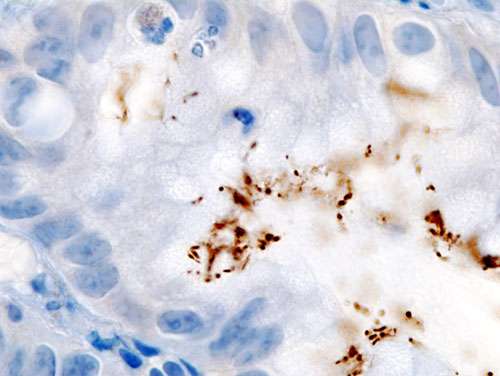
Histopathology of Helicobacter pylori infection in a gastric foveolar pit demonstrated in endoscopic gastric biopsy. Credit: Wikipedia.
Helicobacter pylori infects approximately half of the world's population and is the strongest known risk factor for developing gastric cancer. Gastric cancer is the third most lethal cancer worldwide.
A cluster of genes called the "cag pathogenicity island" renders some strains of H. pylori particularly virulent. However, it is not clear exactly how H. pylori induces gastric cancer.
Now in a study published in the Proceedings of the National Academy of Sciences, Lydia Wroblewski, Ph.D., Richard Peek, MD, and collaborators have shown that in chronically infected mice, carcinogenic strains of H. pylorimobilize a transmembrane protein that marks a distinct population of progenitor cells called Lrig1.
H. pylori was found to stimulate Lrig1-expressing progenitor cells in a cag-dependent manner, and these reprogrammed cells gave rise to a full spectrum of differentiated cells. Moreover, in human samples, Lrig1 expression was enhanced in gastric lesions with premalignant potential.
These findings provide further insight into the detrimental events that develop in response to H. pylori infection.
More information: Lydia E. Wroblewski et al. Targeted mobilization of Lrig1+ gastric epithelial stem cell populations by a carcinogenic Helicobacter pylori type IV secretion system, Proceedings of the National Academy of Sciences (2019). DOI: 10.1073/pnas.1903798116
Journal information: Proceedings of the National Academy of Sciences
Provided by Vanderbilt University