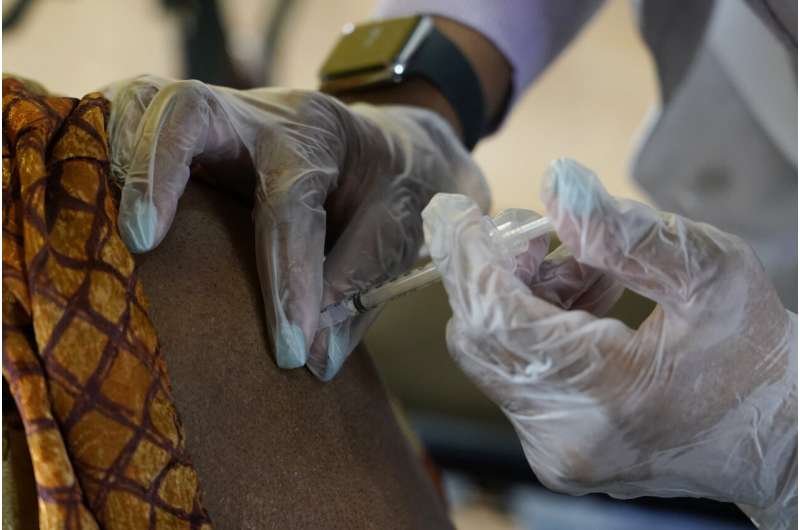
In this Jan. 12, 2021, file photo resident of Harmony Court Assisted Living receives the Pfizer COVID-19 vaccine in Jackson, Miss. The U.S. vaccination drive against COVID-19 stood on the verge of a major new phase as government advisers Thursday, Sept. 23, recommended booster doses of Pfizer's vaccine for millions of older or otherwise vulnerable Americans—despite doubts the extra shots will do much to slow the pandemic. Credit: AP Photo/Rogelio V. Solis, File
The U.S. vaccination drive against COVID-19 stood on the verge of a major new phase as government advisers Thursday recommended booster doses of Pfizer's vaccine for millions of older or otherwise vulnerable Americans—despite doubts the extra shots will do much to slow the pandemic.
Advisers to the Centers for Disease Control and Prevention said boosters should be offered to people 65 and older, nursing home residents and those ages 50 to 64 who have risky underlying health problems. The extra dose would be given once they are at least six months past their last Pfizer shot.
Deciding who else might get one was far tougher. While there is little evidence that younger people are in danger of waning immunity, the panel offered the option of a booster for those 18 to 49 who have chronic health problems and want one.
But the advisers refused to go further and open boosters to otherwise healthy front-line health care workers who aren't at risk of severe illness but want to avoid even a mild infection.
"We might as well just say give it to everyone 18 and older. We have a very effective vaccine and it's like saying, 'It's not working.' It is working," said Dr. Pablo Sanchez of Ohio State University, who helped block the broadest booster option.
Still, getting the unvaccinated their first shots remains the top priority, and the panel wrestled with whether the booster debate was distracting from that goal.
All three of the COVID-19 vaccines used in the U.S. still are highly protective against severe illness, hospitalization and death, even amid the spread of the extra-contagious delta variant. But only about 182 million Americans are fully vaccinated, just 55% of the population.
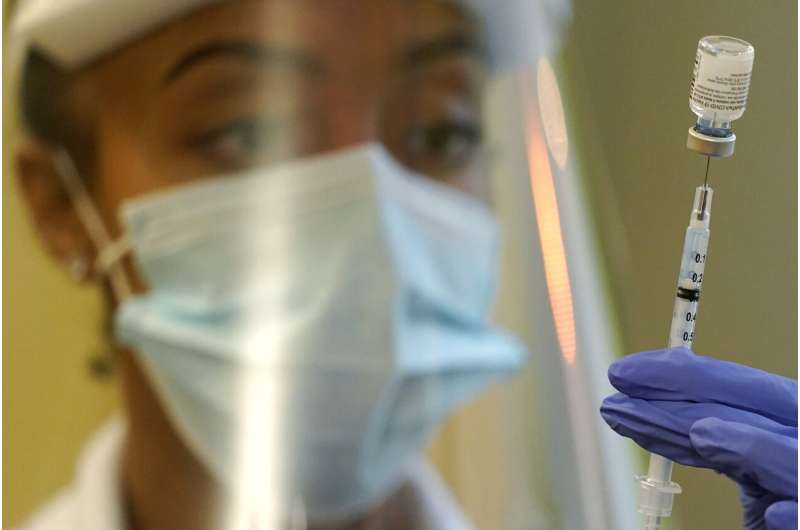
In this Jan. 8, 2021, file photo, a pharmacist prepares a syringe of the Pfizer vaccine for COVID-19, at Queen Anne Healthcare, a skilled nursing and rehabilitation facility in Seattle. The U.S. vaccination drive against COVID-19 stood on the verge of a major new phase as government advisers Thursday, Sept. 23, recommended booster doses of Pfizer's vaccine for millions of older or otherwise vulnerable Americans—despite doubts the extra shots will do much to slow the pandemic. Credit: AP Photo/Ted S. Warren, File
"We can give boosters to people, but that's not really the answer to this pandemic," said Dr. Helen Keipp Talbot of Vanderbilt University. "Hospitals are full because people are not vaccinated. We are declining care to people who deserve care because we are full of unvaccinated COVID-positive patients."
Thursday's decision represented a dramatic scaling back of the Biden administration plan, announced last month, to dispense boosters to nearly everyone to shore up their protection. Late Wednesday, the Food and Drug Administration, like the CDC, signed off on Pfizer boosters for a much more targeted slice of the American population than the White House envisioned.
It falls to the CDC to set final U.S. policy on who qualifies for the extra shot. The CDC usually follows its advisers' recommendations. A final decision from the agency was expected later Thursday.
The booster plan marks an important shift in the nation's vaccination drive. Britain and Israel are already giving a third round of shots over strong objections from the World Health Organization that poor countries don't have enough for their initial doses.
CDC Director Dr. Rochelle Walensky opened Thursday's meeting by stressing that vaccinating the unvaccinated remains the top goal "here in America and around the world."
Walensky acknowledged that the data on who really needs a booster right away "are not perfect." "Yet collectively they form a picture for us," she said, "and they are what we have in this moment to make a decision about the next stage in this pandemic."
The CDC panel stressed its recommendations will be changed if new evidence shows more people need a booster.
In this May 21, 2021, file photo, licensed vocational nurse Angeline Gabuten administers the Pfizer COVID-19 vaccine to a patient at Providence Edwards Lifesciences vaccination site in Santa Ana, Calif. The U.S. vaccination drive against COVID-19 stood on the verge of a major new phase as government advisers Thursday, Sept. 23, recommended booster doses of Pfizer's vaccine for millions of older or otherwise vulnerable Americans—despite doubts the extra shots will do much to slow the pandemic. Credit: AP Photo/Jae C. Hong, File
The CDC advisers expressed concern over the millions more Americans who received Moderna or Johnson & Johnson shots early in the vaccine rollout. The government still hasn't considered boosters for those brands and has no data on whether it's safe or effective to mix-and-match and give those people a Pfizer shot.
"I just don't understand how later this afternoon we can say to people 65 and older you're at risk for severe illness and death but only half of you can protect yourselves right now," said Dr. Sarah Long of Drexel University.
About 26 million Americans got their last Pfizer dose at least six months ago, about half of whom are 65 or older. It's not clear how many more would meet the CDC panel's initial booster qualifications.
CDC data shows the vaccines still offer strong protection for all ages, but there is a slight drop among the oldest adults. And immunity against milder infection appears to be waning months after people's initial immunization.
For most people, if you're not in a group recommended for a booster, "it's really because we think you're well-protected," said Dr. Matthew Daley of Kaiser Permanente Colorado. "This isn't about who deserves a booster, but who needs a booster."
Among people who stand to benefit from a booster, there are few risks, the CDC concluded. Serious side effects from the first two Pfizer doses are exceedingly rare, including heart inflammation that sometimes occurs in younger men. Data from Israel, which has given nearly 3 million people—mostly 60 and older—a third Pfizer dose, has uncovered no red flags.
The panelists also wrestled with how to even tell when a booster is needed. While an extra dose revs up numbers of virus-fighting antibodies, those naturally wane over time and no one knows how long the antibody boost from a third Pfizer dose will last—or how much protection it really adds, since the immune system also forms additional defenses after vaccination.
The U.S. has already authorized third doses of the Pfizer and Moderna vaccines for certain people with weakened immune systems, such as cancer patients and transplant recipients. Other Americans, healthy or not, have managed to get boosters, in some cases simply by asking.
© 2021 The Associated Press. All rights reserved. This material may not be published, broadcast, rewritten or redistributed without permission.