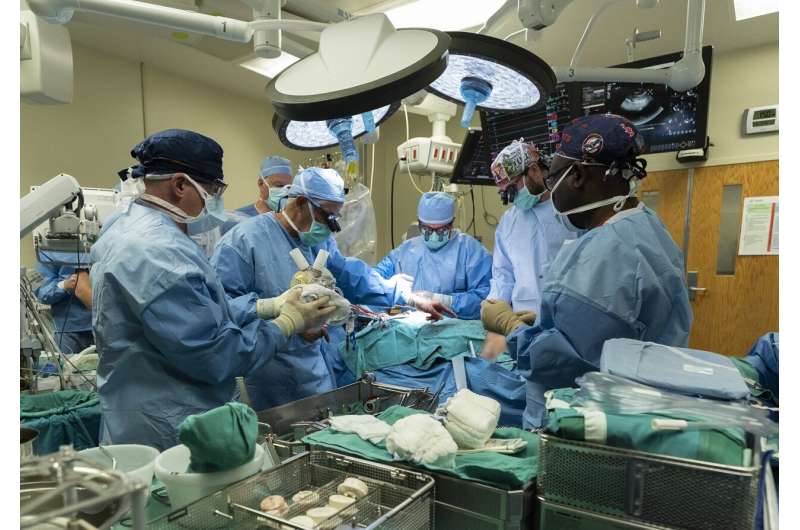
Cardiothoracic surgeons with UofL Health – Jewish Hospital and the University of Louisville. Credit: UofL Health
A cardiothoracic surgical team with UofL Health—Jewish Hospital and the University of Louisville has performed the world's first Aeson bioprosthetic total artificial heart implantation in a female patient. The investigational device, currently intended as a bridge to heart transplant, is part of an Early Feasibility Study (EFS) sponsored by CARMAT, a French medical device company, in partnership with UofL, UofL Health—Jewish Hospital and the UofL Health—Trager Transplant Center.
Led by cardiothoracic surgeons Mark Slaughter, M.D., and Siddharth Pahwa, M.D., both of UofL Health—UofL Physicians and the UofL School of Medicine, the team performed the implant of the device on Sept. 14, 2021 at UofL Health—Jewish Hospital. The same team completed the nation's second implantation in a male patient last month, also at Jewish Hospital.
"For the other half of the world's population, completion of this procedure by the Jewish Hospital team brings new hope for extended life," said Slaughter, UofL Health surgical director of heart transplant and professor and chair of the Department of Cardiovascular and Thoracic Surgery in the UofL School of Medicine. "Size limitations can make it harder to implant artificial hearts in women, but the Aeson artificial heart is compact enough to fit inside the smaller chest cavities more frequently found in women, which gives hope to a wider variety of men and women waiting for a heart transplant and increases the chances for success."
More than 3,500 individuals are awaiting a heart transplant in the U.S. and 900 of them are women. There are few treatment options for patients with biventricular heart disease, meaning both the left and right sides of the heart are not pumping blood adequately. The Aeson device is designed to solve the limitations of current left-ventricular assist devices (LVAD), which pump blood in just one chamber, by pumping blood in both heart chambers. Aeson also contains pressure sensors that estimate the patient's blood pressure and automatically adapts cardiac output according to the sensor information. It is fully implanted as a heart replacement and powered by a portable external power supply.
During this procedure, the Aeson total artificial heart was implanted into a 57-year-old Kentucky woman with severe biventricular heart failure during an eight-hour surgery. The recipient, whose identity is being withheld upon request, was referred to the Advanced Heart Failure Therapies Program at Jewish Hospital earlier this year with end-stage heart failure and had undergone cardiac surgery years before. The patient is recovering well in the cardiovascular intensive care unit (CVICU). Jewish Hospital is just one of four programs in the nation approved to perform this clinical trial procedure.
"The varying pumping ability of the Aeson device increases its viability among more patients," said Pahwa, UofL Physicians cardiothoracic surgeon and assistant professor in the UofL Department of Cardiovascular and Thoracic Surgery. "While other devices are set at a fixed rate or create a continuous flow, CARMAT has developed the Aeson to automatically adjust the flow, creating an improved performance to meet the body's changing blood flow needs."
Currently, the Aeson artificial heart is tested as a bridge to transplant for patients with end-stage biventricular heart failure, allowing more time for the patient to receive a permanent heart organ transplant. The device already has been approved for such use in Europe, where approximately 20 devices have been implanted. It currently is being tested in the U.S. as part of a feasibility study approved by the Food and Drug Administration. The first Aeson artificial heart in North America was implanted in a male patient in July at Duke University Medical Center. The second implantation, also in a male patient, was performed at Jewish Hospital in August. This third North American implantation is the first to involve a female patient.
"Even as we have fought this deadly pandemic, our researchers and health care providers have also been on the front lines of improving care and quality of life for not only Kentuckians, but for people around the world," said Kentucky Gov. Andy Beshear. "I am proud that UofL, Jewish Hospital and their doctors are leading the world in implanting this promising and innovative device that could offer hope and time to thousands of people, including our wives, mothers and other loved ones, in coming years."
Stéphane Piat, chief executive officer of CARMAT, said, "This third implant in the U.S. was a landmark event not only because it allowed us to finalize the enrollment of the first cohort of patients of the EFS, but very importantly because it is the first time ever that our device has helped a woman suffering from heart failure. This achievement confirms that the size limitations for adults are minimal, which makes us very confident in Aeson's potential to become a therapy of choice for a broad patient population."
Preclinical research for CARMAT's artificial heart began at UofL more than five years ago. Researchers at UofL's Cardiovascular Innovation Institute (CII) tested Aeson's autoregulation capability, which allows the device to adapt its flows according to the patient's needs by detecting changes of pressure in the device. UofL researchers have conducted preclinical testing of artificial heart components and mechanical assist devices at CII for many years, testing some portion of nearly every mechanical assist device that is commercially available today.
"This world-first artificial heart implant into a female patient is another demonstration of UofL Health's commitment to provide both the world-class care of today and develop the world-class standards of tomorrow," said John Walsh, chief administrative officer of Jewish Hospital. "We celebrate this first as a milestone and recognize the hard work of Drs. Slaughter and Pahwa and the entire team. The true impact of their work will be measured in the dozens, hundreds and thousands of lives improved in the years to come."
The patient who received the nation's second Aeson implant, on Aug. 20, 2021, continues to improve at Jewish Hospital. An update is expected in the coming weeks.
Provided by University of Louisville