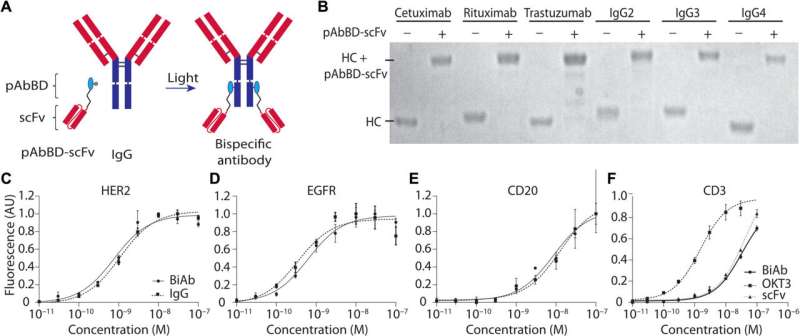
BiAb production mediated by a pAbBD results in six different, pure constructs that bind their respective tumor targets as expected. (A) An anti-CD3 scFv was fused with a pAbBD. Two hours of irradiation with nondamaging long-wavelength ultraviolet (UV) light induces covalent attachment of the fusion protein to the IgG Fc region. (B) Six human antibodies—rituximab, cetuximab, trastuzumab, IgG2, IgG3, and IgG4—and the resulting BiAbs produced by photocrosslinking with pAbBD–anti-CD3 fusion protein were analyzed on a reducing SDS–polyacrylamide gel electrophoresis (SDS-PAGE). Unbound, excess pAbBD-scFv was removed via filtration. The bands represent IgG heavy chains (HC) before and after photocrosslinking. (C to E) HER2+ T617, epidermal growth factor receptor–positive (EGFR+) MDA-MB-468, and CD20+ HT1080 cell lines were seeded, fixed, and blocked with normal goat serum. Binding curves of photocrosslinked (C) anti-HER2 (trastuzumab) × anti-CD3, (D) anti-EGFR (cetuximab) × anti-CD3, and (E) anti-CD20 (rituximab) × anti-CD3 BiAbs and the respective monospecific antibodies from which they were derived were obtained after incubation with a fluorescent secondary antibody. Fluorescence intensity was measured using a plate reader. The dissociation constant (Kd) values were (A) 0.9 nM for the BiAb and 1.16 nM for monoclonal antibody, (B) 0.60 nM for the BiAb and 0.49 nM for the monoclonal antibody, and (C) 8.2 nM for the BiAb and 12 nM for the monoclonal antibody. All coefficient of determination (R2) values are greater than 0.9. (F) Human T cells were incubated with serial dilutions of anti-EGFR × anti-CD3 scFv BiAb, free anti-CD3 scFv, and positive control OKT3, and binding was measured via flow cytometry. The Kd values were found to be 1.89, 35, and 34 nM for OKT3, CD3 scFv, and BiAb with R2 values of 0.99, 0.985, and 0.99, respectively. AU, arbitrary units. Credit: Science Advances (2022). DOI: 10.1126/sciadv.abn4613
One of the reasons that cancer is notoriously difficult to treat is that it can look very different for each patient. As a result, most targeted therapies only work for a fraction of cancer patients. In many cases, patients will have tumors with no known markers that can be targeted, creating an incredible challenge in identifying effective treatments. A new study seeks to address this problem with the development of a simple methodology to help differentiate tumors from healthy, normal tissues.
This new study, published in Science Advances, was led by Andrew Tsourkas, Professor in Bioengineering and Co-Director of the Center for Targeted Therapeutics and Translational Nanomedicine (CT3N), who had what he describes as a "crazy idea" to use a patient's antibodies to find and treat their own tumors, taking advantage of the immune system's innate ability to identify tumors as foreign. This study, spearheaded by Burcin Altun, a former postdoctoral researcher in Tsourkas's lab, and continued and completed by Fabiana Zappala, a former graduate student in Penn Bioengineering, details their new method for site-specifically labeling "off-the-shelf" and native serum autoantibodies with T cell–redirecting domains.
Researchers have known for some time that cancer patients will generate an antibody response to their own tumors. These anti-tumor antibodies are quite sophisticated in their ability to specifically identify cancer cells; however, they are not sufficiently potent to confer a therapeutic effect. In this study, Tsourkas's team converted these antibodies into bispecific antibodies, thereby increasing their potency. T cell-redirecting bispecific antibodies are a new form of targeted therapeutic that forms a bridge between tumor cells and T cells which have been found to be as much as a thousand-times more potent than antibodies alone. By combining the specificity of a patient's own antibodies with the potency of bispecific antibodies, researchers can effectively create a truly personalized therapeutic that is effective against tumors.
In order to test out this new targeted therapeutic approach, the Tsourkas lab had to develop an entirely new technology, allowing them to precisely label antibodies with T cell targeting domains, creating a highly homogeneous product. Previously it has not been possible to convert native antibodies into bispecific antibodies, but Tsourkas's Targeted Imaging Therapeutics and Nanomedicine or TITAN lab specializes in the creation of novel targeted imaging and therapeutic agents for detection and treatment of various diseases. "Much is yet to be done before this could be considered a practical clinical approach," says Tsourkas. "But I hope at the very least this works stimulates new ideas in the way we think about personalized medicine."
In their next phase, Tsourkas's team will be working to separate anti-tumor antibodies from other antibodies found in patients' serum (which could potentially redirect the bispecific antibodies to other locations in the body), as well as examining possible adverse reactions or unintended effects and immunogenicity caused by the treatment. However, this study is just the beginning of a promising new targeted therapeutic approach to cancer treatment.
More information: Fabiana Zappala et al, Rapid, site-specific labeling of "off-the-shelf" and native serum autoantibodies with T cell–redirecting domains, Science Advances (2022). DOI: 10.1126/sciadv.abn4613
Journal information: Science Advances
Provided by University of Pennsylvania