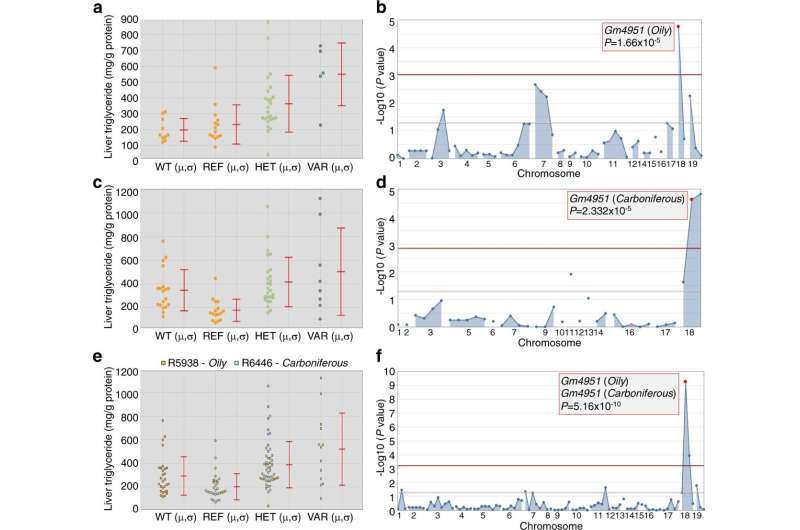
Identification and mapping of Oily and Carboniferous. a Liver triglyceride data from G3 mice plotted vs. genotype at the Oily mutation site of Gm4951 after four weeks on a HFD (n = 10 mice in WT; n = 14 mice in REF; n = 25 mice in HET; n = 5 mice in VAR). b Manhattan plot showing P values calculated using a likehood-ratio test from an additive model of inheritance. c Liver triglyceride data from G3 mice plotted vs. genotype at the Carboniferous mutation site of Gm4951 after four weeks on a HFD (n = 19 mice in WT; n = 16 mice in REF; n = 26 mice in HET; n = 8 mice in VAR). d Manhattan plot showing P values calculated using a likehood-ratio test from an additive model of inheritance. e, f Liver triglyceride data from pedigrees R5938 (Oily) and R6446 (Carboniferous) were pooled together in superpedigree analysis and plotted as in a and c (n = 29 mice in WT; n = 30 mice in REF; n = 51 mice in HET; n = 13 mice in VAR); Manhattan plot as in (b) and (d). P values were calculated using a likehood-ratio test from an additive model of inheritance. WT, C57BL/6 J mice age-matched with G3 mice; REF, G3 mice homozygous for the reference allele of the indicated gene; HET, G3 mice heterozygous for the reference allele and for the mutant allele; VAR, G3 mice homozygous for the mutant allele. Each data point represents one mouse; mean (μ) and SD (σ) are indicated (a, c, e). Data are from one experiment. Credit: Nature Communications (2022). DOI: 10.1038/s41467-022-31812-4
Using a genetic screening platform developed by a UT Southwestern Nobel Laureate, scientists with the Center for the Genetics of Host Defense at UT Southwestern Medical Center have identified genetic mutations that contribute to nonalcoholic fatty liver disease (NAFLD), providing a potential future target for therapeutic interventions.
While obesity and diabetes are well-known risk factors for fatty liver disease, the UT Southwestern researchers were able to identify a new cause of fatty liver disease absent obesity—a reduction in the level of Predicted gene 4951 (Gm4951), which in turn leads to non-alcoholic fatty liver disease. Currently there are no approved medications or treatments for the disease, which is rapidly emerging as a major cause of chronic liver disease in the United States. Identifying the role of this gene in the development of the disease provides an important new direction for those studying the disease to find potential treatments.
"We identified a rare non-obese mouse model of NAFLD caused by GM4951 deficiency. This study lays the groundwork for the future development of approaches to activate the human Gm4951 homolog to combat NAFLD," said lead author Zhao Zhang, Ph.D., Assistant Professor in the Center for the Genetics of Host Defense and the Division of Endocrinology in Internal Medicine. The findings appear in Nature Communications.
The Center for the Genetics of Host Defense is directed by Bruce Beutler, Ph.D., a Regental Professor and one of four Nobel Laureates at UT Southwestern. Dr. Beutler received the 2011 Nobel Prize in Physiology or Medicine for discovering an important family of receptors that allow mammals to sense infections when they occur, triggering a powerful inflammatory response. Dr. Beutler also developed the largest mouse mutagenesis program in the world, along with a forward genetic screen platform that allows researchers in the Center to screen for more than half of all genes in the mouse genome. Along with a means of instantly identifying mutations responsible for both quantitative and qualitative phenotypes, the program allows for rapid discovery of many new components of the immune system.
"This study identified a potential human homolog of mouse GM4951 and the interaction of GM4951 with another human NAFLD/NASH associated protein HSD17B13, suggesting the discovery is likely conserved in humans," said Dr. Zhang, whose lab is working to understand the molecular mechanism of metabolic diseases, with the underlying aim of translating this knowledge into novel therapeutic strategies.
The current study is built on the forward genetic screen platform, which allowed the researchers to identify two semi-dominant allelic missense mutations (Oily and Carboniferous) of Gm4951 and define a critical role for GTPase-mediated translocation in hepatic lipid metabolism. Among their findings, the researchers found that loss of GM4951 causes NAFLD without obesity, that GM4951 promotes lipid oxidation to prevent lipid from accumulation in the liver and that GM4951 functions as a GTPase to translocate HSD17B13 to lipid droplets.
GM4951 is a poorly characterized protein and this study defined the role of GM4951 as a GTPase involved in lipid oxidation, said Dr. Zhang. The GM4951-deficient mice developed nonalcoholic fatty liver disease on a high-fat diet with no changes in body weight or glucose metabolism, the researchers noted.
An estimated one-quarter of adults in the U.S. have nonalcoholic fatty liver disease (NAFLD), an excess of fat in liver cells that can cause chronic inflammation and liver damage, increasing the risk of liver cancer, liver failure and need for transplant. NAFLD has become the most common cause of liver disease worldwide.
In recent decades, it has been suggested that changes in the lifestyles clearly drive the risk in the prevalence of NAFLD. However, hepatic fat content varies substantially among individuals with equivalent adiposity, indicating that genetic factors contribute to the development of NAFLD. Dr. Zhang's lab is investigating more than 20 genes in which mutations affect liver triglyceride with no change in body weight to identify new mechanisms of NAFLD.
More information: Zhao Zhang et al, Loss of immunity-related GTPase GM4951 leads to nonalcoholic fatty liver disease without obesity, Nature Communications (2022). DOI: 10.1038/s41467-022-31812-4
Journal information: Nature Communications
Provided by UT Southwestern Medical Center