A newly developed antibody targeting a signalling pathway that is frequently active in solid tumours has shown encouraging signs of efficacy in its first trial in humans, researchers will report at the 24th EORTC-NCI-AACR Symposium on Molecular Targets and Cancer Therapeutics in Dublin, Ireland, today (Wednesday).
A patient with advanced malignant melanoma has shown signs of tumour shrinkage and has been receiving treatment for more than 30 weeks without any serious adverse side-effects. Other patients with advanced non-small cell lung cancer (NSCLC), mesothelioma, kidney cancer and biliary tract cancer also have had extended treatments with stable disease.
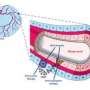
The compound is a monoclonal antibody known as RG7212. It targets a soluble protein in the blood called TWEAK (Tumour Necrosis Factor (TNF)-like weak inducer of apoptosis. TWEAK binds to the receptor Fn14 (fibroblast growth factor-inducible 14) on the cell membrane, and this is the signal for a cascade of subsequent events, including activation of multiple cancer-promoting pathways. One objective of this phase I trial was to determine whether blocking of TWEAK-Fn14 signalling with RG7212 would reduce activation of these pathways.
Dr Ulrik Lassen (MD, PhD), head of the oncology phase I unit at the Rigshospitalet, Copenhagen, Denmark, will tell the Symposium: "TWEAK and Fn14 are a pair of cell signalling molecules that are part of the large family of tumour necrosis factors that play a number of roles in the development of cancer. The Fn14-receptor is over-expressed in a variety of advanced solid tumours, and signalling through TWEAK-Fn14 enhances multiple processes associated with malignancy.
"Preclinical data have shown that the RG7212 monoclonal antibody successfully targets and blocks the action of TWEAK and that it works most effectively in tumour cells where there is increased Fn14 expression. Therefore, we expected that using RG7212 to block TWEAK signalling in patients whose tumours express Fn14 might prevent cancer cells from growing and proliferating."
Fn14 is over-expressed in at least 30% of several solid tumours, including pancreatic cancer, breast cancer, non-small cell lung cancer and malignant melanoma.
From July 2011, Dr Lassen and his colleagues recruited 38 patients with treatment-refractory solid tumours to the phase I trial. All patients had tumours expressing the Fn14 receptor. They received an intravenous dose of the drug either once weekly or once every three weeks, with a dose range of 200 to 3600 mg.
Dr Lassen said: "Several of the patients continue to receive study treatments, and encouraging signs of clinical benefit have already been observed. A patient with heavily pre-treated, metastatic melanoma with no mutation in the BRAF gene has shown evidence of tumour regression when scanned using computerised tomography (CT) and remains on study after more than 30 weeks of treatment. Four other patients have had partial metabolic responses confirmed by positron emission tomography (PET) scans.
"Prolonged stable disease has been seen in several patients. Overall, 11 of the 38 patients (29%) have received more than 12 weeks of study treatment, with several receiving 18 or more weeks of RG7212 therapy, including those with refractory NSCLC, melanoma, mesothelioma, breast cancer, renal cell carcinoma, and biliary tract cancer.
"We found that RG7212 has an excellent safety profile across a broad dose range on each schedule. We saw no dose-limiting toxicities and no patient discontinued study treatments for treatment-related adverse side-effects. The phase I data show that RG7212 is quite safe for multi-cycle administration in patients with advanced cancer. Results from tests of blood and tumour samples suggest that it would be feasible to administer the drug over a prolonged period of time.
"There are encouraging pharmacodynamic data (effects of the drug on tumour and blood samples), including durable inhibition of TWEAK, inhibition of signalling pathways controlled by Fn14, and inhibition of tumour cell proliferation."
Study investigators from four leading cancer research centres in Denmark, The Netherlands and Canada are recruiting patients to the trial, particularly those with advanced metastatic melanoma expressing Fn14. "These results are encouraging and support additional studies of RG7212 in combination with other treatments," said Dr Lassen.
Professor Stefan Sleijfer, the scientific chair of the EORTC-NCI-AACR Symposium, from Erasmus University Medical Centre (The Netherlands), commented: "Fn14 is a novel target for treatment in cancer. In view of these data, this drug certainly deserves further studies in Fn14 expressing tumours."
More information: The study is funded by F. Hoffmann-La Roche, Inc.
Provided by ECCO-the European CanCer Organisation