In 10% of human tumors there is a family history of hereditary disease associated with mutations in identified genes. The best examples are the cases of polyps in the large intestine associated with the APC gene and breast cancer associated with BRCA1 and BRCA2 genes. In the remaining 90% of cases are believed to have an increased risk of developing cancer in relation to genetic variants less powerful but more often, for example, doubles the risk of having a tumor that lacks this small change, called polymorphism.
In the last decade, hundreds of studies have been conducted looking for polymorphisms associated with a greater propensity to suffer some of the most frequent human tumors. These tests, called GWAS, have found a common problem: many times the tiny genetic change observed appears to have no activity or function to explain because it is associated with more cancer.
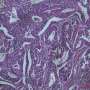
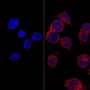
Today, an article published in Cell Reports led by Manel Esteller, Director of Epigenetics and Cancer Biology, Bellvitge Biomedical Research Institute (IDIBELL) , ICREA researcher and Professor of Genetics at the University of Barcelona, solves part of this enigma . Research shows that in one in four human tumor exist genetic polymorphisms associated with increased risk of cancer that cause epigenetic changes that modifies the expression of neighboring genes.
"This work represents a Herculean effort to analyze by bioinformatics a lot of biological information. To be exact, we have studied a million genetic changes in relation to half a million epigenetic changes in 3,500 tumors representing the thirteen most common cancers, including breast, lung and colon" says Manel Esteller.
"We have seen in 25% genetic polymorphisms associated with increased cases of cancer, but not directly alter the gene, which was caused a change in DNA methylation; ie the gene was switched off without breaking it. These results add epigenetic changes as another key factor to consider in the inheritance of cancer and the contribution of the familial component to the disease" concludes Esteller.
More information: Heyn H, Sayols S, Moutinho C, Vidal E, Sanchez-Mut JV, Stefansson OA, Nadal E, Moran S, Eyfjord JE, Gonzalez-Suarez E, Pujana MA, Esteller M. Linkage of DNA Methylation Quantitative Trait Loci to Human Cancer Risk. Cell Reports, DOI: 10.1016/j.celrep.2014.03.016, 2014.
Journal information: Cell Reports
Provided by IDIBELL-Bellvitge Biomedical Research Institute