What if a failed leukemia drug could reduce the risk of Alzheimer's disease? A team at the University of Kentucky recently led an effort to investigate this hypothesis. Their results were published today in the journal, Human Molecular Genetics.
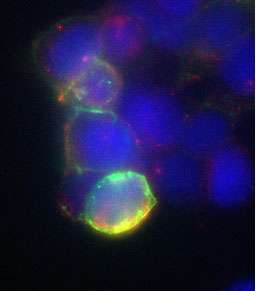
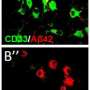
The UK researchers, led by Steve Estus at the Sanders-Brown Center on Aging, study a genetic variant in a gene called CD33 that reduces the risk of Alzheimer's disease. The Estus group recently published findings suggesting that this variant promotes production of a truncated form of the CD33 protein that lacks a putative functional domain. The CD33 protein is present on microglia, the resident immune cells of the brain. CD33 is thought to dull the microglial response and thereby inhibit clearance of toxic debris that builds up in the Alzheimer's brain.
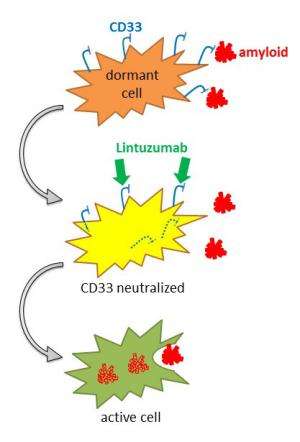
Based on their genetic data, Manasi Malik of the Estus lab, hypothesized that a CD33 inhibitor might activate sluggish microglia to reduce the risk of Alzheimer's disease even beyond the protection provided by the CD33 genetic variant. The group may have found such an inhibitor in Lintuzumab, a CD33 antibody that was tested in clinical trials for acute myeloid leukemia.
Although the drug was ineffective at killing leukemic cells and is currently not on the market, the researchers found that Lintuzumab effectively eliminates CD33 from the surface of immune cells. Next, they plan to see if immune cells treated with Lintuzumab are more effective at clearing the amyloid beta aggregates and dead cells that may lead to symptoms of Alzheimer's. Although more work is needed before human studies, the group's findings overall suggest that a drug that has already been tested safely in humans may prove to be useful against Alzheimer's disease.
The study, which was funded in large part by the National Institute of Aging/National Institutes of Health, included investigators from the UK Departments of Physiology, Internal Medicine, Toxicology and Biostatistics as well as researchers from Pfizer, Inc., the University of Washington, and Mayo Clinic-Jacksonville.
More information: "Genetics of CD33 in Alzheimer's disease and acute myeloid leukemia." Hum. Mol. Genet. first published online March 11, 2015 DOI: 10.1093/hmg/ddv092
Journal information: Human Molecular Genetics
Provided by University of Kentucky