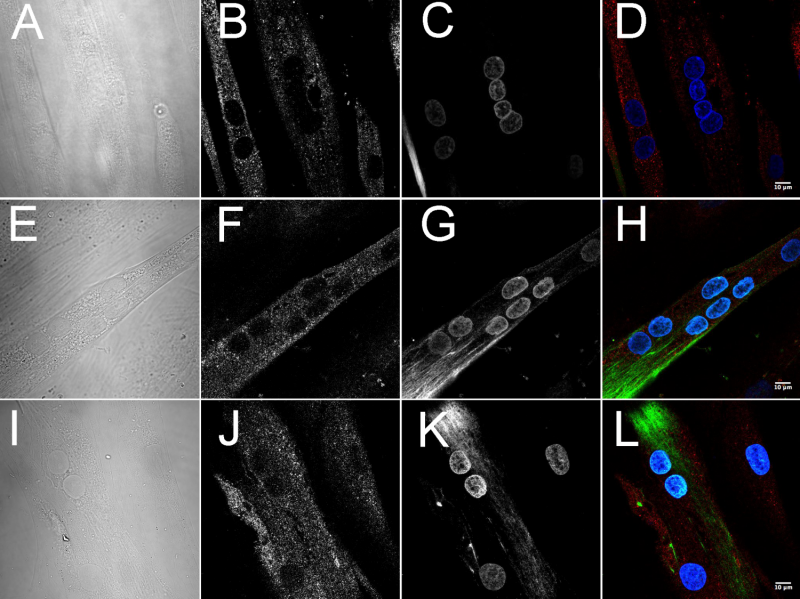
Images of cellular distribution of dystrophin and utrophin in differentiated human myotubes grown on glass coverslips. Panels A-D: quadriceps; E-H: orbicularis oculi; I-L: extraocular muscles. Credit: Sekulic-Jablanovic et al., 2016
In a new study, a research team at Basel University Hospital in Switzerland investigates the biochemical and physiological characteristics of orbicularis oculi, a group of facial muscles that control the eyelids and are selectively spared or involved in different neuromuscular disorders. What they found also helps to explain why another set of muscles—the extraocular muscles that control the movement of the eye—are not affected by Duchenne muscular dystrophy, congenital muscular dystrophy, and aging.
The study, "Functional characterization of orbicularis oculi and extraocular muscles," by Marijana Sekulic-Jablanovic et al., will appear online April 11 ahead of print of the May issue of The Journal of General Physiology (JGP).
Using healthy human muscle cell cultures and differentiated human myotubes grown on glass coverslips, the team led by Susan Treves, PhD and Francesco Zorzato, MD, PhD, of the departments of Anaesthesia and Biomedicine at Basel University Hospital, examined the excitation-contraction coupling (ECC) process and calcium homeostasis in the orbicularis oculi, extraocular muscles, and a third muscle group—the quadriceps.
ECC is the process whereby an electrical signal is converted into a chemical signal leading to muscle contraction. They found that ECC machinery and calcium regulation of human orbicularis oculi are more similar to those of quadriceps than of extraocular muscles.
"Although gene expression in orbicularis oculi is quite different from that in quadriceps, the expression of proteins involved in the ECC process is similar in both cell types," the authors note.
The researchers also gained unique insight into the presence and absence of dystrophin, the protein located primarily in muscles used for movement, and utrophin, which shares both structural and functional similarities with dystrophin.
In patients with neuromuscular disorders, weakness is apparent in the orbicularis oculi. Although mitochondrial myopathies and myasthenia gravis often first target the orbicularis oculi and the extraocular muscles, this is not the case in Duchenne muscular dystrophy.
Duchenne muscular dystrophy (DMD) is the most common muscular dystrophy in children and primarily targets boys, according to the Centers for Disease Control and Prevention, and it results in the loss of the ability to walk between ages 7 and 13 years, and death in the teens or 20s. DMD is caused by a defective gene for dystrophin.
In the study, the researchers found high levels of dystrophin and low levels of utrophin in the quadriceps. Orbicularis oculi and extraocular muscles, however, expressed low levels of dystrophin and high levels of utrophin.
"Our results concerning the expression of utrophin are interesting and most likely explain why ocular and facial muscles are spared in patients with Duchenne muscular dystrophy," the authors say.
The authors note that dystrophin and utrophin may share distinct binding partners since their subcellular distribution, at least in myotubes, is different. Dystrophin has a punctate membrane distribution, whereas utrophin has a patchy more filamentous distribution concentrated within specific subcellular domains. Also promising, dystrophin-deficient mdx mouse studies show that utrophin can functionally compensate in vivo for the lack of dystrophin.
"This supports current strategies aimed at modulating utrophin expression in the therapy for Duchenne muscular dystrophy," the authors conclude. "Taken together, these studies show that satellite cells derived from different muscles are primed and will follow the developmental characteristics of their muscle of origin, a property that can be exploited in laboratories devoted to tissue engineering."
More information: Sekulic-Jablanovic, et al. 2016. J Gen. Physiol. dx.doi.org/10.1085/jgp.201511542
Journal information: Journal of General Physiology
Provided by Rockefeller University Press