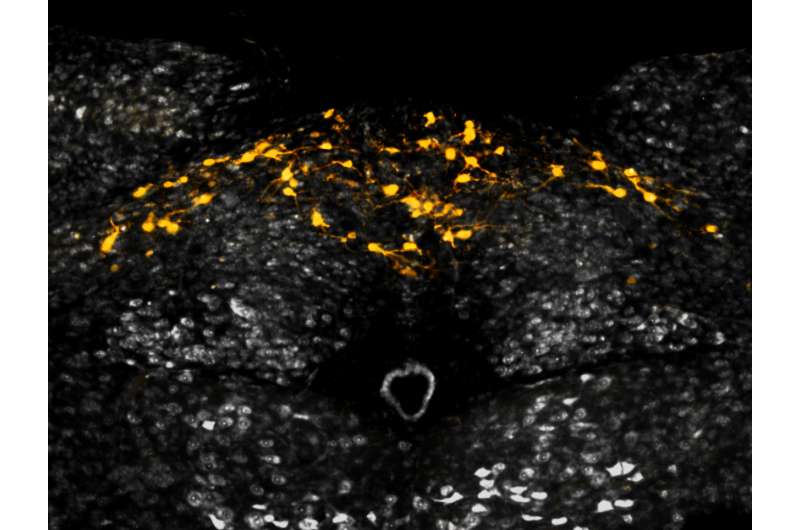
GFRAL-expressing neurons (yellow cells) are found exclusively in the brainstem (pictured), and form the 'emergency circuit' of body weight regulation. Credit: NGM Bio
Scientists at NGM Bio have revealed deep insights into the role that a little-understood human hormone plays in regulating body weight. Named Growth and Differentiation Factor 15 (GDF15), this hormone is typically active only when the body experiences acute or prolonged stress, including following exposure to tissue-damaging toxins, such as chemotherapy, or during chronic disease, such as obesity or cancer. As a result, the GDF15 pathway holds promise for the development of potential therapeutics for diseases of both excess and insufficient body weight.
The paper, published in Nature, uncovers important molecular biology and mechanisms related to this hormone and its receptor, including the crystal structure of the complex. Based on these preclinical findings, NGM and its collaborator, Merck, are advancing multiple drug candidates, including NGM386. NGM386 is an optimized variant of GDF15 that activates its cognate receptor, known as GDNF Receptor Alpha-like (GFRAL), and holds potential as a treatment for obesity, a growing epidemic that affects an estimated 78 million adults in the United States. NGM is also developing NGM120, a monoclonal antibody antagonist of GFRAL that holds potential as a treatment for cachexia. Cachexia is an extreme form of weight loss exacerbated by chemotherapy that afflicts more than half of all cancer patients, for whom it is frequently the proximate cause of death.
"While it had been established that GDF15 is involved in weight-related diseases, the lack of information about its signaling pathway had impeded the development of new medicines harnessing this biology," said Jin-Long Chen, Ph.D., founder and chief scientific officer of NGM Bio. "This publication describes the critical breakthrough that enabled the development of multiple drug candidates for these difficult-to-treat disorders, which could ultimately transform the lives of patients battling obesity and cachexia."
The paper shows that GDF15 binding to GFRAL is required both to protect mice from weight gain in metabolically-stressed conditions and to trigger excessive weight loss in mice treated with chemotherapy. This work also demonstrated that GFRAL forms a complex with the receptor tyrosine kinase RET on the surface of neurons that are localized exclusively in the brainstem, and that these neurons form part of what has been recently dubbed the "emergency circuit" of body weight regulation. This circuit, which resides deep within the brain, lies outside of those areas normally associated with weight control. Until recently, the identity of circulating molecules that could activate this circuit remained elusive. By binding to GFRAL and activating RET, GDF15 'flips the switch' on this circuit, leading to anorexia and weight loss.
NGM's research supports the hypothesis that obesity and cachexia are opposite sides of the same biological process. Drugs such as NGM386 and NGM120 that can toggle the GFRAL 'switch' on or off may have the potential to offer therapeutic benefit for a range of weight-related diseases.
"GFRAL was discovered to be the receptor for GDF15 by one of our dedicated scientists whose commitment to drug discovery prompted her to visit the lab on a holiday weekend to check on an experiment. In the ensuing years, working in collaboration with Merck, our team has advanced multiple product candidates against the GDF15 receptor complex for human testing," said William J. Rieflin, chief executive officer of NGM Bio. "We hope the dedication of our researchers and clinicians will ultimately translate into medicines for patients who struggle with weight-related diseases."
The paper, entitled "Non-homeostatic body weight regulation through a brainstem-restricted receptor for GDF15," published online on Sept. 27 and will appear in the October 12, 2017 print issue of Nature.
More information: Jer-Yuan Hsu et al. Non-homeostatic body weight regulation through a brainstem-restricted receptor for GDF15, Nature (2017). DOI: 10.1038/nature24042
Journal information: Nature
Provided by NGM Bio