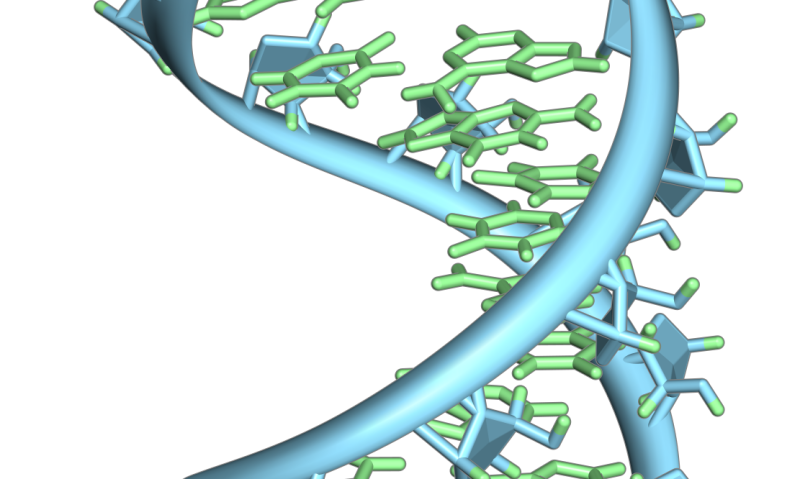
A hairpin loop from a pre-mRNA. Highlighted are the nucleobases (green) and the ribose-phosphate backbone (blue). Note that this is a single strand of RNA that folds back upon itself. Credit: Vossman/ Wikipedia
The RIG-I like receptors (RLRs) are intracellular enzyme sentries that detect viral infection and initiate a first line of antiviral defense. The cellular molecules that activate RLRs in vivo are not clear.
John Karijolich, Ph.D., and colleagues have made the surprising discovery that host-derived RNAs—not viral molecules—activate RLRs. The investigators studied the cellular response to infection with Kaposi's sarcoma-associated herpesvirus (KSHV), an oncogenic virus and AIDS-associated pathogen that causes Kaposi's sarcoma and primary effusion lymphoma (PEL).
In patient-derived PEL cells, the researchers demonstrated that RLRs restrict KSHV lytic reactivation—the viral replication and cell-destroying part of the viral life cycle. They found that RLRs sense host-derived noncoding RNAs, and that an infection-dependent reduction in another host protein results in the accumulation of noncoding RNAs that activate RLRs.
The study, reported in Nature Communications, defines the in vivo substrates of RLRs during an oncogenic virus infection and reveals a relationship between cellular RNA processing and an innate antiviral immune response.
More information: Yang Zhao et al. RIG-I like receptor sensing of host RNAs facilitates the cell-intrinsic immune response to KSHV infection, Nature Communications (2018). DOI: 10.1038/s41467-018-07314-7
Journal information: Nature Communications
Provided by Vanderbilt University