From left: Hanwen Zhu, Ph.D.; Ji Sun, Ph.D.; and Patricia Hixson, Ph.D., all of St. Jude Structural Biology, have determined the detailed structure of an enzyme whose mutated forms are known to cause Parkinson’s disease. Credit: St. Jude Children's Research Hospital
Structural biologists at St. Jude Children's Research Hospital have determined the detailed structure of an enzyme whose mutated forms are known to cause Parkinson's disease. The achievement opens a research pathway to develop new drugs that can specifically target the aberrant form of a protein enzyme called LRRK2.
The researchers, led by Ji Sun, Ph.D., of the St. Jude Department of Structural Biology, published their findings today in the journal Cell.
LRRK2 is a biological enzymatic switch called a kinase. Numerous disease-associated mutations in the gene for LRRK2 produce an over-activated form. This activated enzyme triggers the production of pathological filaments of LRRK2 in brain cells, which causes the cell malfunction and death that disrupts neuronal function in Parkinson's patients.
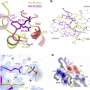
Although other researchers had obtained structures of portions of the large LRRK2 protein, the structure determined by Sun and his colleagues is the first high-resolution structure of the full molecule.
The researchers determined the LRRK2 structures using cryo-electron microscopy, or cryo-EM. This analytical technique involves cooling molecules to super cold temperatures and imaging them using a beam of electrons. By analyzing the patterns of the resulting images, scientists can construct the detailed structure of a molecule.
Obtaining the structure of full-length LRRK2 is critical to designing targeted Parkinson's drugs, Sun said. Only that structure can reveal how the enzyme is regulated, he said. Other researchers had determined the structure of portions of the molecule—including the kinase domain that carries out LRRK2's biochemical switching function. However, regulation of enzymes such as LRRK2 involves other regions of the molecule, which can alter the overall shape of the protein to activate the kinase domain.
"If we only design a drug to target the kinase domain, it's hard to achieve specificity because there are some 600 kinases in the human body, and all the kinases look quite similar," Sun said. "But if you can understand how this protein is regulated, and you target this regulation, you can achieve specificity in designing drugs. So this structure provides a new direction for designing inhibitors of LRRK2."
Importantly, the researchers determined the structure not only of a single LRRK2 molecule, called a "monomer," but also of two linked LRRK2 molecules, called a "dimer." Obtaining the structure of the inactive LRRK2 dimer is important for further studies of LRRK2's function, Sun said.
"In real physiological conditions, LRRK2 is only active when it's in the dimer form," he said. "So this dimer structure gives us a start in piecing together the puzzle of how LRRK2 works in the cell. We are now working on ways to produce an active form of the LRRK2 dimer to study its function."
The researchers' high-resolution structural studies of full-length LRRK2 also pinpointed "disease hotspots" in the molecule. These hotspots are areas of LRRK2 where mutations are known to cause disease. Identifying these hotspots offers important structural clues to how the disease-causing mutations abnormally activate LRRK2, Sun said.
The researchers also obtained the structure of the most prevalent mutant form of LRRK2, called GS2019S, known to cause Parkinson's disease.
"This mutation will be an important drug target, so if we can specifically inactivate this mutant form with a drug, we will have a way to potentially cure this form of the disease," he said.
Sun and his colleagues also used their structural studies to explore an entirely different approach to treating Parkinson's. The researchers discovered that if they genetically altered even a single point of the molecule where the two segments of the dimer are connected, they could abolish the enzyme's ability to produce the pathological LRRK2 filaments that damage and kill brain cells.
"If we could design drugs that do the same thing—prevent the pathological formation of these filaments—that might also be another way to treat Parkinson's disease," Sun said. "This approach has been less well studied than efforts to prevent the mutation-caused gain of function of LRRK2."
More information: Alexander Myasnikov et al, Structural analysis of the full-length human LRRK2, Cell (2021). DOI: 10.1016/j.cell.2021.05.004
Journal information: Cell
Provided by St. Jude Children's Research Hospital