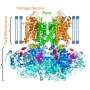
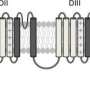
Northwestern Medicine scientists have mapped the complete structure of a voltage-gated sodium channel, proteins in the membrane of cells that play an important role in many diseases. The findings were published in Nature Communications.
"We've known about these proteins for a long time—and essentially what they do—but we didn't know what they look like," said Paul DeCaen, PhD, assistant professor of Pharmacology, who was the corresponding author on the paper. "For pharmacology, you need to know what your target really looks like in order to develop a drug that will fit its shape. Now we have the full picture for one type of sodium channel."
While previous studies had charted certain regions of sodium channels, this is the first complete, and the highest-resolution, structure of a sodium channel to date.
The scientists used crystallography to produce the architecture of a voltage-gated sodium channel from a prokaryotic cell. Leo Ng, PhD, a postdoctoral fellow in DeCaen's lab, was also a co-author of the paper, along with collaborators at Birkbeck College in London.
Voltage-gated sodium channels are responsible for initiating action potential in excitable cells throughout the body, particularly neurons. When the channels don't function properly, and electric signals aren't conducted correctly, the dysfunction can manifest in disorders like epilepsy and some cardiac arrhythmias.
By describing previously unseen components of the sodium channels, the current study provides new insight into their function and a greater understanding of how those proteins select specific ions in order to generate an action potential. The process takes place in an area called the selectivity filter.
"Sodium channels have a difficult job. They only allow sodium ions into the cell despite the presence of a number of other ions, like potassium and calcium," DeCaen explained. "Our collaborators captured a snapshot of these ions as they are passing through, like people crossing the street. Previously, all we had were images when the street was empty."
DeCaen and his collaborators are currently working on understanding the same structure in eukaryotic cells, findings which could eventually lead to a drug template for targeting sodium channels in humans.
"Science is iterative; there are steps and small achievements along the way toward something that has a bigger impact," DeCaen said. "We are accomplishing some small steps right now, but we hope that it's going to become something much bigger."
More information: Altin Sula et al. The complete structure of an activated open sodium channel, Nature Communications (2017). DOI: 10.1038/ncomms14205
Journal information: Nature Communications
Provided by Northwestern University