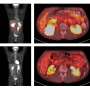
When different types of cells are transplanted with the intent of having them aid in repairing central nervous system (CNS) trauma, what is the fate and function of those cells? A Belgian research team carried out research aimed at answering this question by determining how five varieties of cells - neural stem cells, mouse embryonic fibroblasts, dendritic cells, bone marrow mononuclear cells (BMMNCs) and splenocytes - functioned and survived after transplantation in the CNS.
Their study is published in a recent issue of Cell Transplantation (21:9).
"Severe injuries to the central nervous system due to trauma are among the top five causes of death and represent a significant social and economic health burden on society," said Dr. Peter Ponsaerts of the University of Antwerp. "There are no currently effective treatments to restore damaged or dysfunctional neural tissues, although much progress is expected to come from stem cell research."
Their research investigated how factors such as cell migration, cell graft survival, cell graft immunogenicity and cell graft toxicity are impacted two weeks post-transplantation. Their study examined the fate and function of the five varieties of cells mentioned above after they were transplanted into the CNS of transgenic mice, both healthy and those modeled with CNS injury with subsequent inflammation.
"We did not see a significant increase for any cell types in cell migration under inflammatory conditions when compared to healthy conditions," said the authors. "With regard to cell graft survival, to our surprise, only a few of the cells were able to survive under both healthy and inflammatory conditions."
However, they found "striking differences" in how endogenous immune responses against grafted cells were organized. They speculated that differences were due to both the degree of microglia and astrocyte invasion. Responses of both were observed in each different cell variety. BMMNC and splenocyte grafts were less invaded by microglia, but displayed high levels of astrocyte invasion.
Finally, with regard to cell graft toxicity, the researchers found that "none of the cell populations could directly contribute to remyelination." They speculated that two weeks might not be sufficient time for proper differentiation of grafted NSCs, although endogenous remyelination does occur within a two-week time period.
"Our results warrant further research to better understand – and eventually control – cell graft induced tissue damage and activation of the brain's innate immune system," they wrote.
More information: Praet, J.; Reekmans, K.; Lin, D.; De Vocht, N.; Bergwerf, I.; Tambuyzer, B.; Daans, J.; Hens, N.; Goossens, H.; Pauwels, P.; Berneman, Z.; Van der Linden, A.; Ponsaerts, P. Cell Type-Associated Differences in Migration, Survival, and Immunogenicity Following Grafting in CNS Tissue. Cell Transplant. 21(9):1867-1881; 2012.
Journal information: Cell Transplantation