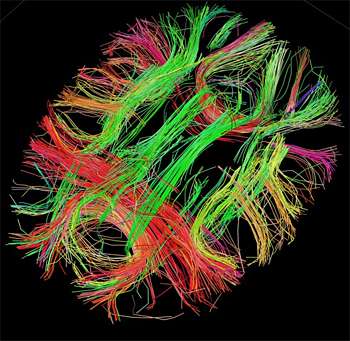
White matter fiber architecture of the brain. Credit: Human Connectome Project.
(Medical Xpress)—Neurologists have known that an intrinsic connection exists between the consolidation of memory and neural activity during sleep. Indeed, over the course of the last century, researchers have increasingly characterized sleep as a brain state optimizing memory consolidation; an earlier emphasis on rapid-eye-movement (REM) sleep has shifted in recent years toward the function of slow-wave sleep (SWS) as an active brain state—for instance, involving the transfer of long-term fear memory from the hippocampus into cortical structures.
A group of researchers from the Max Planck Institute of Experimental Medicine and Ludwig Maximilian University recently published the results of study that explored the function of two transcription factors that modulate the circadian cycle in an attempt to establish a connection between memory and sleep. They have published their findings in the Proceedings of the National Academy of Sciences.
For the experiment, the researchers bred mice lacking the transcription factors SHARP1 and SHARP2, which are known to control the circadian cycle. They also modulate a pair of core clock factors implicated in homeostatic sleep, neuronal plasticity and working memory. The mice lacking both transcription factors showed enhanced cortex-dependent remote fear memory formation and improved reversal learning—tasks associated with the anterior cingulate cortex—but had no alteration in hippocampus-dependent recent fear memory formation.
Two signaling pathways for hippocampal learning that interest researchers are mitogen-activated protein kinase (MAPK), which is known to be involved with processes of long-term memory formation. The other is insulin-related growth factor 2 (IGF2). The researchers discovered that in the double-knockout mutant mice, cortex-dependent learning tasks were enhanced, paralleled with elevated IGF2 expression and MAPK signaling in the cortex, but not in the hippocampus. They conclude that the elevated IGF2 expression in the anterior cingulate cortex activates MAPK signaling to enhance memory consolidation in cortex-dependent learning tasks. From this, the researchers conclude that the control of sleep and memory consolidation share multiple common molecular mechanisms.
Previous studies have demonstrated that IGF2 appears to be a potent enhancer of memory in the hippocampus; the current study also establishes its importance to memory function in the cortex. Such factors are putative therapeutical agents for age-related cognitive decline and neurodegenerative diseases.
The researchers write, "We could show that IGF2 expression and MAPK activation display a circadian regulation in the cortex of wild type mice that appears to be uncoupled upon loss of the negative clock modulators SHARP1/2. Our study thus paves the way to investigate circadian and sleep-associated aspects of cortex-dependent memory consolidation in more depth in the future."
In addition, the researchers found that as they aged, the mutant mice lacking SHARP1/2 displayed marked declines in cognitive performance. It's unclear if this was caused by the elevated IGF2 expression or the lack of SHARP1/2 functions, and this result points toward future research pathways to understand neural metabolism and age-related cognitive decline.
More information: "Enhanced memory consolidation in mice lacking the circadian modulators Sharp1 and -2 caused by elevated Igf2 signaling in the cortex." PNAS 2015 ; published ahead of print June 22, 2015, DOI: 10.1073/pnas.1423989112
Abstract
The bHLH transcription factors SHARP1 and SHARP2 are partially redundant modulators of the circadian system. SHARP1/DEC2 has been shown to control sleep length in humans and sleep architecture is also altered in double mutant mice (S1/2−/−). Because of the importance of sleep for memory consolidation, we investigated the role of SHARP1 and SHARP2 in cognitive processing. S1/2−/− mice show enhanced cortex (Cx)-dependent remote fear memory formation as well as improved reversal learning, but do not display alterations in hippocampus (Hi)-dependent recent fear memory formation. SHARP1 and SHARP2 single null mutants do not display any cognitive phenotype supporting functional redundancy of both factors. Molecular and biochemical analyses revealed elevated insulin-related growth factor 2 (IGF2) signaling and increased phosphorylation of MAPK and S6 in the Cx but not the Hi of S1/2−/− mice. No changes were detected in single mutants. Moreover, adeno-associated virus type 2-mediated IGF2 overexpression in the anterior cingulate cortex enhanced remote fear memory formation and the analysis of forebrain-specific double null mutants of the Insulin and IGF1 receptors revealed their essential function for memory formation. Impaired fear memory formation in aged S1/2−/− mice indicates that elevated IGF2 signaling in the long term, however, has a negative impact on cognitive processing. In summary, we conclude that the bHLH transcription factors SHARP1 and SHARP2 are involved in cognitive processing by controlling Igf2 expression and associated signaling cascades. Our analyses provide evidence that the control of sleep and memory consolidation may share common molecular mechanisms.
Journal information: Proceedings of the National Academy of Sciences
© 2015 Medical Xpress