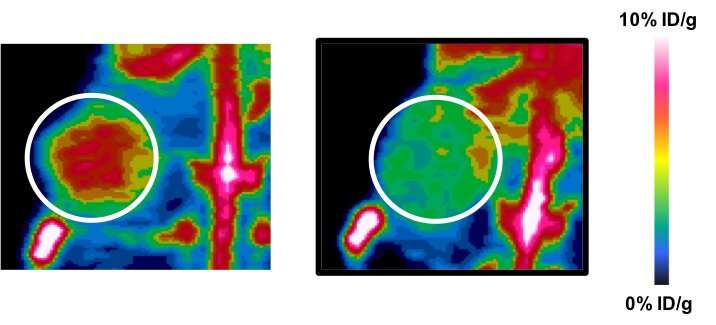
Left, illuminated tumor by iPET expressing immunotherapy target, compared to control tumor (right). Credit: UTSW
A novel imaging test shows promise for identifying kidney cancer patients most likely to benefit from immunotherapy.
In a study published today in the Journal for ImmunoTherapy of Cancer, investigators with the UT Southwestern Medical Center Kidney Cancer Program developed a new test to illuminate kidney cancers that may respond to checkpoint inhibitors.
The strategy involved transforming an immunotherapy drug, atezolizumab (Tecentriq, Genentech/Roche Group), into a diagnostic tracer. Atezolizumab, which is used to treat lung, breast, and bladder cancer, binds to and disables PD-L1, a protein that cancer cells display on their surface to shut off approaching killer immune cells. By labeling atezolizumab with zirconium-89 (Zr89), a radioactive metal generated using a cyclotron, the investigators were able to visualize atezolizumab using PET (positron emission tomography). As such, a single, very small dose of Zr89-atezolizumab can be used to evaluate whether tumors deploy PD-L1 to suppress immune cells and whether drugs disabling this pathway may be effective.
Currently, immunotherapy drugs benefit less than 50 percent of kidney cancer patients. With immuno-PET, or iPET, as a screening tool, the investigators hope to identify those patients who will benefit. Marking the first time this type of theranostic (drug turned into a diagnostic test) is deployed for kidney cancer, the approach opens a molecular window to what is going on inside a cancer patient.
In proof-of-principle experiments, a team led by Dr. James Brugarolas, one of the corresponding authors of the study and the Director of the UT Southwestern Kidney Cancer Program, showed that Zr89-atezolizumab was able to illuminate kidney tumors with high levels of PD-L1. As part of the study, investigators selected tumors from two patients, one with high PD-L1 and another with low PD-L1, and transplanted them into mice. The mice were then injected with Zr89-atezolizumab intravenously and evaluated by PET. As predicted from the mouse studies, the patient with the high PD-L1 tumor had substantial regression of his metastases when treated with nivolumab (Opdivo, Bristol-Myers Squibb), which targets the PD-L1 pathway.
"The development of tests predicting which patients respond to immunotherapy is critical," said Dr. Hans Hammers, an immunotherapy expert with the Kidney Cancer Program.
Zr89-atezolizumab was filed with the U.S. Food and Drug Administration by the Cyclotron and Radiochemistry Program led by Dr. Xiankai Sun at UT Southwestern, also a corresponding author of the study, and is now proceeding to evaluation in patients in a clinical trial at UT Southwestern's Harold C. Simmons Comprehensive Cancer Center.
The clinical trial is made possible through a $600,000 translational award to Dr. Brugarolas' team by the V Foundation for Cancer Research. Support for the preclinical studies was provided through a Specialized Program of Research Excellence (SPORE) grant from the National Cancer Institute.
"We are hopeful that iPET will identify kidney cancer patients most likely to benefit from checkpoint inhibitors," said Dr. Alex Bowman, Principal Investigator in the clinical trial, along with Dr. Brugarolas and Dr. Orhan Öz.
A second trial also is planned at the Simmons Cancer Center using Zr89-atezolizumab to evaluate the impact of stereotactic body radiation therapy (SBRT) on PD-L1 expression in kidney cancer patients.
"SBRT has the potential to induce inflammation and activate an immune response, and we are excited to evaluate this therapy further in patients using iPET," said Dr. Raquibul Hannan, a leader in radiation therapy for kidney cancer and Principal Investigator of the study. The SBRT trial will be funded by the Kidney Cancer Congressionally Directed Medical Research Program.
More information: Joseph Vento et al, PD-L1 detection using 89Zr-atezolizumab immuno-PET in renal cell carcinoma tumorgrafts from a patient with favorable nivolumab response, Journal for ImmunoTherapy of Cancer (2019). DOI: 10.1186/s40425-019-0607-z
Provided by UT Southwestern Medical Center