Credit: CC0 Public Domain
Scientists from Russia and Italy studied a new axis of the pathway that prevents the development of liver fibrosis. The role of GILZ protein in curbing the disease progression was shown in a study using mice models and confirmed by clinical data. These findings can be used in the treatment of liver fibrosis in humans. The research was published in the journal Cell Death & Disease.
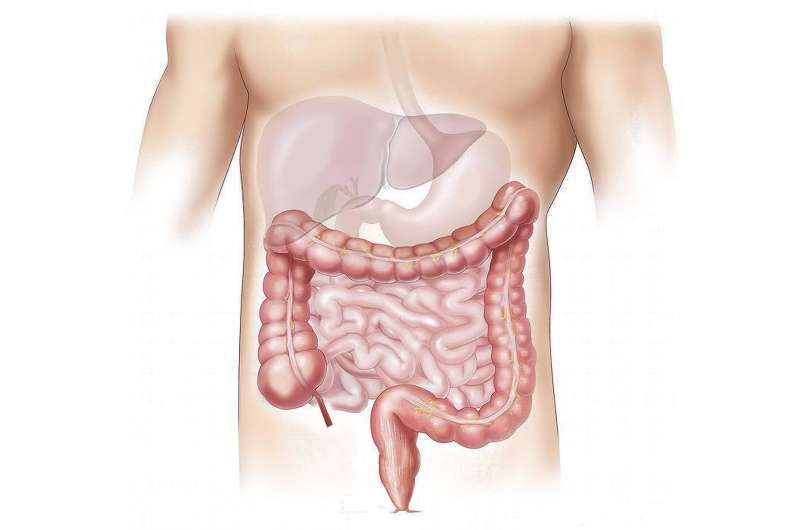
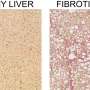
Fibrosis combines an overgrowth of connective tissue and a decline in the liver function that can be caused by a viral infection, alcohol intoxication, autoimmune diseases or other liver disorders. If left untreated, fibrosis can lead to cirrhosis and even death. Inflammatory processes—complex cascades of molecular interactions between the cells of the immune system—play an important role in the progression of fibrosis, therefore, its treatment requires thorough understanding of those processes at the molecular level. The most common anti-inflammatory agents, such as glucocorticoids, are widely used in the treatment of autoimmune diseases and other problems, however, in the case of liver fibrosis, they are likely to cause severe side effects.
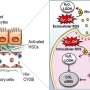
In their recent study, researchers from Skoltech, the University of Perugia, and the University of Florence (Italy) focused on the GILZ protein. GILZ expression leads to changes in cellular processes similar to those triggered by glucocorticoids. The team experimented with a model of liver fibrosis induced in GILZ knockout mice and observed rapid progression of the disease. The scientists verified their hypothesis about the effect of GILZ on the progression of fibrosis using gene expression data on patients with liver fibrosis and obtained evidence of lower GILZ levels in those patients. Downregulation of the upstream CCR2 protein restored resistance to the progression of liver fibrosis.
The team's findings suggest that GILZ is a promising anti-hepatic fibrosis drug target.
"Importantly, there is a strong correlation between our data on mice and clinical data on humans, which is rarely the case with lab results obtained using model objects and even mammals that may never be confirmed in humans. Now we have every reason to expect that by controlling the signaling pathway which involves GILZ, one could treat inflammatory liver diseases in humans," professor Timofei Zatsepin of the Skoltech Center for Life Sciences (CLS) explains.
More information: Sara Flamini et al. Glucocorticoid-induced leucine zipper regulates liver fibrosis by suppressing CCL2-mediated leukocyte recruitment, Cell Death & Disease (2021). DOI: 10.1038/s41419-021-03704-w
Provided by Skolkovo Institute of Science and Technology