Cytoglobin: Key player in preventing liver disease
Researchers have discovered that the use of Cytoglobin (CYGB) as an intravenous drug could delay liver fibrosis progression in mice.
CYGB, discovered in 2001 by Professor Norifumi Kawada, is present in hepatic stellate cells, the cells that produce fibrotic molecules such as collagens when the liver has acute or chronic inflammation induced by different etiologies. The enhancement of CYGB on these cells or the injection of recombinant CYGB has the effect of suppressing liver damage and cirrhosis.
These findings were published in the February 2021 issue of the journal Hepatology.
Anti-fibrotic therapy remains an unmet medical need in human chronic liver diseases.
A research team led by Professor Norifumi Kawada, Osaka City University (OCU), reported the novel anti-fibrotic properties of CYGB, a respiratory protein expressed in hepatic stellate cells (HSCs), the main cell type involved in liver fibrosis. In mice with advanced liver fibrosis, both enhancement of CYGB or recombinant CYGB injection can suppress hepatocyte damage and liver fibrosis. In chimera mice with a human liver, the injected CYGB did not show any adverse side effects.
"Fixing the liver after injury is a highly orchestrated, coordinated process, and inhibiting the fibrosis could return the liver to a healthy condition," said Norifumi Kawada, M.D., Ph.D., Dean of OCU Medical School and senior author of the study.
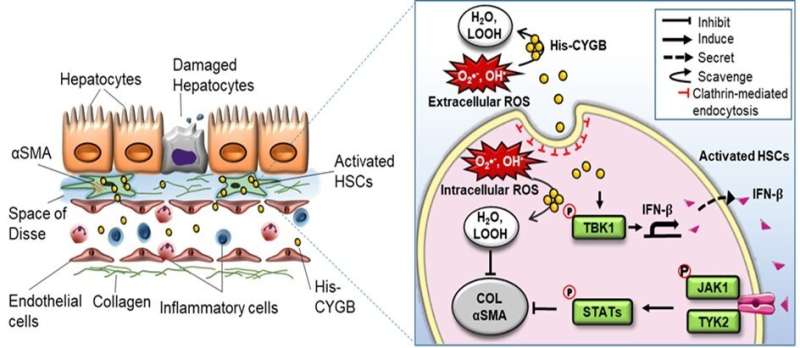
Liver injury starts with hepatocyte damage, following several conditions including inflammatory cell infiltration, activation of HSCs, and the production of harmful reactive oxygen species (ROSs).
Dr. Kawada, along with Le Thi Thanh Thuy, Ph.D., OCU scientist, and their colleagues, observed that when they culture human HSCs under recombinant human CYGB treatment, it can enter the cellular organelles, hunt the harmful ROSs, and prevent the activation of HSCs leading to inhibit collagen production.
To identify potential treatment strategies, Dr. Kawada and his team developed several animal models of liver injury and investigated how the liver responds to CYGB regulation and treatment.
In both mouse models of bile flow obstruction induced cholestasis and high fat diet induced fatty liver disease, the absence of CYGB made the liver injury more severe, however, when CYGB was enhanced, the liver injury subsided.
In the next set of experiments, Dr. Kawada and his group tried to generate two other mouse models of advanced liver fibrosis using different chemical agents and applied recombinant human CYGB protein by intravenous injection. Interestingly, the therapeutic protein dramatically suppressed liver injuries, inflammation and fibrosis without any side effects.
Using the results of this research as a foothold, the research team hopes to start clinical trials using this new anti-fibrotic therapy.
More information: Ninh Quoc Dat et al. 6His‐tagged Recombinant Human Cytoglobin Deactivates Hepatic Stellate Cells and Inhibits Liver Fibrosis by Scavenging Reactive Oxygen Species, Hepatology (2021). DOI: 10.1002/hep.31752