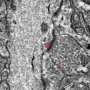
Super-resolution microscopy reveals the presence of active release sites (green) on dopamine neurons (purple). Credit: Kaeser lab
Studied for decades for its critical role in movement control and reward-seeking behaviors, the neurotransmitter dopamine has been the focus of numerous efforts to understand its activity, particularly when it goes awry in disorders such as Parkinson's and addiction.
While researchers have made great strides, less is known about the mechanisms that healthy dopamine cells use to release the neurotransmitter, a gap that has limited scientists' ability to develop treatments for a range of dopamine-related conditions.
Now, researchers from Harvard Medical School have for the first time identified the molecular machinery responsible for the precise secretion of dopamine in the brain.
Their work, published online in Cell on Feb. 1, identifies specialized sites in dopamine-producing neurons that release the dopamine in a fast, spatially precise manner—a finding that runs counter to current models of how the neurotransmitter transmits signals in the brain.
"The dopamine system plays an essential role in many diseases, but fewer studies have asked the fundamental question of how healthy dopamine neurons release the neurotransmitter," said senior study author Pascal Kaeser, assistant professor of neurobiology at Harvard Medical School.
"If your car breaks down and you want it fixed, you want your mechanic to know how a car works," he added. "Similarly, a better understanding of dopamine in the laboratory could have a tremendous impact on the ability to treat disorders in which dopamine signaling goes awry in the long term."
In the brain, roughly 0.01 percent of neurons are responsible for dopamine production, but they control a broad and diverse range of brain processes, including motor control, the reward system, learning and memory.
Dopamine research has centered on its dysfunction and on the protein receptors that neurons use to receive dopamine, said Kaeser. Despite the neurotransmitter's importance, studies on how it is released in the brain under normal circumstances have been limited, he added.
Promiscuous No More
To identify the molecular machinery responsible for dopamine secretion, Kaeser and his colleagues focused on dopamine-producing neurons in the midbrain, which are involved in the neural circuitry underlying movement and reward seeking.
They first searched for active zones—specialized neurotransmitter release sites located at synapses, the junctions that connect one neuron to the another. Using super-resolution microscopy to image sections of the brain into which dopamine neurons project, the team found that dopamine neurons contained proteins that mark the presence of active zones.
These zones indicate that a neuron may engage in fast synaptic transmission, in which a neurotransmitter signal is precisely transferred from one neuron to another within milliseconds.
This was the first evidence of fast active zones in dopamine neurons, which were previously thought to engage in only so-called volume transmission—a process in which the neurotransmitter signals slowly and nonspecifically across relatively large areas of the brain.
Active zones were found at lower densities in dopamine neurons than in other neurons, and additional experiments revealed in detail how the neurotransmitter is rapidly secreted and reabsorbed at these sites.
"I think that our findings will change how we think about dopamine," Kaeser said. "Our data suggest that dopamine is released in very specific locations, with incredible spatial precision and speed, whereas before it was thought that dopamine was slowly and promiscuously secreted."
In another set of experiments, the researchers used genetic tools to delete several active zone proteins. Deleting one specific protein, RIM, was sufficient to almost entirely abolish dopamine secretion in mice. RIM has been implicated in a range of diseases including neuropsychiatric and developmental disorders.
Deleting another active zone protein, however, had little or no effect on dopamine release, suggesting that dopamine secretion relies on unique specialized machinery, the authors said.
"Our study indicates that dopamine signaling is much more organized than previously thought," said study first author Changliang Liu, an Alice and Joseph Brooks Postdoctoral Fellow and a Gordon Fellow in the Kaeser lab.
"We showed that active zones and RIM, which is associated with diseases such as schizophrenia and autism spectrum disorders in human genetic studies, are key for dopamine signaling," Liu said. "These newly identified mechanisms may be related to these disorders and may lead to new therapeutic strategies in the future."
The team is now working to investigate these active zones in greater detail to build a deeper understanding of their role in dopamine signaling and how to manipulate them.
"We are deeply invested in learning the entire dopamine signaling machine. Right now, most treatments supply the brain with dopamine in excess, which comes with many side effects because it activates processes that shouldn't be active," Kaeser said.
"Our long-term hope is to identify proteins that only mediate dopamine secretion," he said. "One can imagine that by manipulating the release of dopamine, we may be better able to reconstruct normal signaling in the brain."
Journal information: Cell
Provided by Harvard Medical School


.jpg)