Credit: National Jewish Health
Researchers at National Jewish Health and around the world report today that a three-drug combination of medications can improve lung function, reduce exacerbations and improve quality of life for people with cystic fibrosis who have at least one copy of the most common cystic fibrosis mutation, Phe508del. Based in part on these findings, the U.S. Food and Drug Administration recently approved the combination, elexacaftor/tezacaftor/ivacaftor, known as Trikafta, that will ultimately help 90 percent of people with cystic fibrosis.
"This news is the best we have had in my career for people with cystic fibrosis," said Jennifer Taylor-Cousar, MD, MSCS, co-director of the Adult Cystic Fibrosis Program at National Jewish Health and co-author on papers simultaneously published today in The New England Journal of Medicine and The Lancet. "We are finally closing in on a dream that was launched 30 years ago when the gene for cystic fibrosis was discovered."
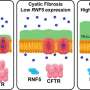
Cystic fibrosis results from a genetic mutation in the CFTR gene, which produces a faulty ion channel that prevents chloride and other ions from exiting cells in the lungs and other organs. With chloride unable to exit cells, so water cannot follow it, thick mucus builds up in the lungs and gastrointestinal organs. The thick mucus provides a favorable environment for bacterial and other infections to develop. Patients suffer repeated infections of the lungs, which result in lung damage over time and eventually premature death.
In recent years, several new medications have been developed that directly impact the faulty CFTR ion channel. One kind, known as a corrector, helps correct misfolding of the protein, which prevents it from reaching the cell surface. Another kind, known as a potentiator, helps it function better once it reaches the cell surface. The medications approved in 2018, double combinations of one corrector and one potentiator, helped people with two copies of the phe508del mutation, about 50 percent of people with cystic fibrosis.
The new combination adds a second corrector to the previously approved combination. As reported today, it helped people with one copy of phe508del and worked much better than the two-drug combination for patients with two copies of phe508del. Lung function, as measured by the volume of air expelled in one second, improved 13.8 percent for people with one copy of phe508del and an additional 10 percent on top of the previously approved double combination for people with two copies of F508del who took the three-drug combination.
Acute worsening of symptoms, known as exacerbations, dropped 63 percent for these people with cystic fibrosis. Exacerbations, most often triggered by viral respiratory infections, are the most common cause of lung damage that eventually leads to respiratory failure. By reducing exacerbations, the triple-combination medication holds the promise of extending patients' lives.
"While I am incredibly excited by the new options we now have for treating cystic fibrosis, we have more work to do," said Dr. Taylor-Cousar. "We need to find effective treatments for the remaining 10 percent of patients who do not have a phe508del mutation. Even more, we need to find a cure for this disease."
Journal information: New England Journal of Medicine , The Lancet
Provided by National Jewish Health