Copper folds protein into precursors of Parkinson's plaques
Researchers at North Carolina State University have figured out how copper induces misfolding in the protein associated with Parkinson's disease, leading to creation of the fibrillar plaques which characterize the disease. This finding has implications for both the study of Parkinson's progression, as well as for future treatments.
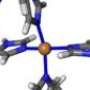
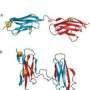
The protein in question, alpha-synuclein, is the major component of fibrillar plaques found in Parkinson's patients. Researchers had already discovered that certain metals, including copper, could increase the rate of misfolding by binding with the protein, but were unsure of the mechanism by which this binding took place.
"We knew that the copper was interacting with a certain section of the protein, but we didn't have a model for what was happening on the atomic level," says Frisco Rose, Ph.D. candidate in physics and lead author of the paper describing the research. "Think of a huge swing set, with kids all swinging and holding hands – that's the protein. Copper is a kid who wants a swing. There are a number of ways that copper could grab a swing, or bind to the protein, and each of those ways would affect all of the other kids on the swing set differently. We wanted to find the specific binding process that leads to misfolding."
Rose and NC State colleagues Dr. Miroslav Hodak, research assistant professor of physics, and Dr. Jerzy Bernholc, Drexel Professor of Physics and Director of the Center for High Performance Simulation, developed a series of computer simulations designed to ferret out the most likely binding scenario.
According to Hodak, "We simulated the interactions of hundreds of thousands of atoms, which required multiple hundred thousand CPU-hour runs to study the onset of misfolding and the dynamics of the partially misfolded structures."
The number of calculations was so large that Hodak and Bernholc had to devise a new method to make it possible for a computer to process them. Only supercomputers like Jaguar, Oak Ridge National Laboratory's most powerful supercomputer – the most powerful in the United States, in fact – were up to the task. But the simulations finally revealed the binding configuration most likely to result in misfolding.
Their results appear in the June 14 edition of Nature Scientific Reports.
The researchers hope that their finding will advance our understanding of Parkinson's, one of the most common – and devastating – neurological diseases. "Understanding the molecular mechanism of Parkinson's disease should help researchers in developing drugs that treat the disease rather than merely alleviate symptoms," Bernholc says.
More information: "Mechanism of copper(II)-induced misfolding of Parkinson's disease protein" Frisco Rose, Miroslav Hodak, Jerzy Bernholc, Nature Scientific Reports, June 14, 2011.
Abstract
α-synuclein (αS) is a natively unfolded pre-synaptic protein found in all Parkinson's disease patients as the major component of fibrillar plaques. Metal ions, and especially Cu(II), have been demonstrated to accelerate aggregation of αS into fibrillar plaques, the precursors to Lewy bodies. In this work, copper binding to αS is investigated by a combination of quantum and molecular mechanics simulations. Starting from the experimentally observed attachment site, several optimized structures of Cu-binding geometries are examined. The most energetically favorable attachment results in significant allosteric changes, making αS more susceptible to misfolding. Indeed, an inverse kinematics investigation of the configuration space uncovers a dynamically stable β-sheet conformation of Cu-αS that serves as a nucleation point for a second β-strand. Based on these findings, we propose an atomistic mechanism of copper-induced misfolding of αS as an initial event in the formation of Lewy bodies and thus in PD pathogenesis.