June 21, 2011 report
New study finds HIV Achilles Heel
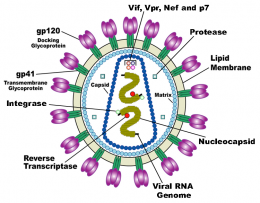
(Medical Xpress) -- A new study published in the Proceedings of the National Academy of Sciences shows how scientists have used a mathematical tool to possibly identify an Achilles heel in HIV which may lead to new vaccines and treatments.
One of the main things that make HIV so difficult to combat is its extreme ability to mutate. However, this research has identified what are being called HIV sectors and are groups of amino acids that are rarely seen making mutations. The researchers believe that the virus needs to maintain these sectors in order to survive and it is these sectors that should be the main target of treatments.
Through a joint enterprise combining the Massachusetts Institute of Technology, Harvard, University and Massachusetts General Hospital, the group called the Ragon Institute is behind the study. The Ragon Institute was established in 2009 to bring scientists together for the study in HIV/AIDS and other similar diseases.
The lead researchers were not biologists but rather specialized in chemistry. Professor of chemistry and chemical engineering at MIT Arup Chakraborty partnered with assistant professor of chemistry from the Universite Pierre et Marie Curie Vincent Dahirel. The two chemists joined forces with a long-time HIV researcher and director of the Ragon Institute Bruce Walker.
Using a 1950s statistical method known as random matrix theory, Chakraborty and Dahirel looked at the HIV proteins and sequences taken from a huge database of HIV patients trying to determine which segment was least able to tolerate mutations. What they found was sector three on the HIV protein known as Gag.
It was determined that part of this sector was responsible for the outer edges of the honeycomb structure that makes up the internal shell of the virus. If this structure suffered too many mutations, it would collapse.
Dr. Walker has spent years studying the rare HIV patients known as “elite controllers.” These patients are able to control their HIV virus with their own immune system and no medication. What they found was the main target of these patient’s immune system was this same sector three.
The main hypothesis determined from this study was that new vaccines and treatments should not focus on a random attack but a focused attack on sector three. Professor of Medicine at Harvard and fellow Ragon colleague Dan Barouch plans to test this hypothesis in monkeys.
More information: Coordinate linkage of HIV evolution reveals regions of immunological vulnerability, PNAS, Published online before print June 20, 2011, doi: 10.1073/pnas.1105315108
Abstract
Cellular immune control of HIV is mediated, in part, by induction of single amino acid mutations that reduce viral fitness, but compensatory mutations limit this effect. Here, we sought to determine if higher order constraints on viral evolution exist, because some coordinately linked combinations of mutations may hurt viability. Immune targeting of multiple sites in such a multidimensionally conserved region might render the virus particularly vulnerable, because viable escape pathways would be greatly restricted. We analyzed available HIV sequences using a method from physics to reveal distinct groups of amino acids whose mutations are collectively coordinated (“HIV sectors”). From the standpoint of mutations at individual sites, one such group in Gag is as conserved as other collectively coevolving groups of sites in Gag. However, it exhibits higher order conservation indicating constraints on the viability of viral strains with multiple mutations. Mapping amino acids from this group onto protein structures shows that combined mutations likely destabilize multiprotein structural interactions critical for viral function. Persons who durably control HIV without medications preferentially target the sector in Gag predicted to be most vulnerable. By sequencing circulating viruses from these individuals, we find that individual mutations occur with similar frequency in this sector as in other targeted Gag sectors. However, multiple mutations within this sector are very rare, indicating previously unrecognized multidimensional constraints on HIV evolution. Targeting such regions with higher order evolutionary constraints provides a novel approach to immunogen design for a vaccine against HIV and other rapidly mutating viruses.
© 2010 PhysOrg.com