July 23, 2012 report
Study finds breast cancer cells able to turn off interferon production to avoid immune response
(Medical Xpress) -- Researchers working in Australia have found that certain breast cancer cells are able to switch off the gene that is responsible for causing the production of interferon, an immunity response protein that the body uses to fight off viral and bacterial infections. Because of this, the team writes in their paper published in Nature Medicine, cancer cells are able to spread to other parts of the body, particularly bone, without being attacked by the immune system.
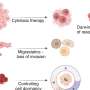
One of the big problems with cancers of all kinds, and particularly breast cancer, is that it spreads from one part of the body to another. No one ever dies from breast cancer directly, it’s only when it spreads to vital organs that it becomes deadly. Thus, a lot of research has gone into finding out how cancers spread and more importantly, how to stop them.
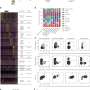
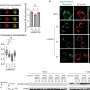
In this new research, to get a better idea of what goes on when cancer spreads from the breast to other parts of the body, the team collected tissue samples from breast cancer patients as well as from dissections of mice. In studying the samples the team found that the cancer cells were able to switch off a gene called IRF7 in people and mice whose cancers spread to other parts of the body. IRF7 is responsible for inciting the production of interferon when infections are detected. Repressing the production of interferon allows the cancer cells to easily spread to bone marrow because it prevents an immunity response. Thus, the cells are able to mask their presence and hide from their attackers.
To counter this effect, the team tried two approaches. In the first they tried putting the gene back into the cells in a way that didn’t allow them to be switched off by the cancer cells. The second way involved injecting test mice with interferon. The team reports that both methods were effective in preventing the spread of the cancer cells, though they urge caution about speculating that any new kind of miracle drug therapy is imminent. They say a lot more testing needs to be done before any patients receive such treatment.
Some patients already receive interferon injections for other ailments, such as some skin cancers, hepatitis and even HIV treatment. Thus far though, no patients with breast cancer have been treated with the immunity response protein.
More information: Silencing of Irf7 pathways in breast cancer cells promotes bone metastasis through immune escape, Nature Medicine (2012) doi:10.1038/nm.2830
Abstract
Breast cancer metastasis is a key determinant of long-term patient survival. By comparing the transcriptomes of primary and metastatic tumor cells in a mouse model of spontaneous bone metastasis, we found that a substantial number of genes suppressed in bone metastases are targets of the interferon regulatory factor Irf7. Restoration of Irf7 in tumor cells or administration of interferon led to reduced bone metastases and prolonged survival time. In mice deficient in the interferon (IFN) receptor or in natural killer (NK) and CD8+ T cell responses, metastasis was accelerated, indicating that Irf7-driven suppression of metastasis was reliant on IFN signaling to host immune cells. We confirmed the clinical relevance of these findings in over 800 patients in which high expression of Irf7-regulated genes in primary tumors was associated with prolonged bone metastasis–free survival. This gene signature may identify patients that could benefit from IFN-based therapies. Thus, we have identified an innate immune pathway intrinsic to breast cancer cells, the suppression of which restricts immunosurveillance to enable metastasis.
© 2012 Medical Xpress