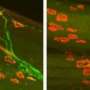
Reconnecting nerves to their target muscles
European researchers are working toward restoration of mobility in neuromuscular disease and trauma. Using miniature scaffolds to guide nerve regeneration, they are seeking to ensure proper functional connections between peripheral nerves and their target muscles.
Peripheral nerves are those located outside the central nervous system (CNS) (the brain and spinal chord). They are like insulated electrical cables whose final connection points must correspond to the signals received by each peripheral nerve from within the CNS.
Thus, peripheral nerves send the signal to specific muscles to either contract or relax. The muscles in doing so then move the bones to which they are connected in logical and useful ways.
In the case of tissue damage to the neuromuscular system (NMS) due to disease or trauma, peripheral nerve lesions result in an inability to transmit signals to the muscles.
Corresponding efforts toward nerve regeneration have fallen short given the difficulty of organised re-enervation leading to restoration of original function.
In other words, progress has been made in regenerating nervous tissue but it is still difficult to regenerate it in a way that forms functionally relevant neuromuscular junctions. Like an old-fashioned switchboard, if the incoming call (nervous signal) is not connected to the appropriate person (specific muscle fibres) the message is not transmitted properly.
Carbon nanotubes (CNTs), literally nano-scale tubes of carbon, are promising potential tissue scaffolds and may be just what the NMS needs.
European researchers initiated the ‘Biocompatability of carbon nanoparticles with tissues of the neuromuscular system’ (NMS-CNT) to build a long-term European consortium working toward the use of carbon nanoparticles in tissue repair of the NMS.
Scientists evaluated a variety of configurations including single-walled CNTs (SWCNTs), multi-walled CNTs (MWCNTs) and ultra-long single-walled CNTs (ULSWCNTs) in particular with respect to solubility.
Ongoing research is directed at enhancing the solubility of ULSWCNTs. These are particularly promising candidates for guided nerve regeneration given their longer length and relatively straight geometry. In addition, investigators are carrying out cytotoxicity tests as well as in vivo testing in animal models.
Completion of the project should provide the scientific foundations for development of CNT-based guided tissue regeneration in the NMS and eventual restoration of mobility in thousands of people with nervous system diseases or traumatic injuries.