Protein that represses critical checkpoint protein for cellular growth helps drive tumor development
(Medical Xpress)—One of the hallmarks of cancer is unchecked cellular growth. Fortunately, our cells contain a number of tumor suppressor proteins, including the cell cycle regulator p21, to keep cell growth in check. The protection conferred by p21, however, can be overridden by an overactive histone-modifying enzyme called PRMT6. This protein represses p21 expression, thereby promoting tumor growth and preventing senescence in breast cancer cells, A*STAR scientists have found.
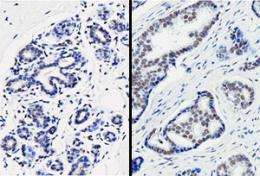
Elevated expression levels of PRMT6 had been reported in a number of cancer types, including breast, cervix, bladder, prostate and lung cancer. However, the protein's role in malignant growth had not been well established. To elucidate PRMT6's function, cancer biologist Ernesto Guccione and his co-workers at the A*STAR Institute of Molecular and Cell Biology in Singapore turned to breast cancer cell lines in which PRMT6 is overexpressed when compared with normal tissue (see image). The researchers used chromatin immunoprecipitation assays, which can determine whether a given protein binds to a specific genomic sequence, and showed that PRMT6 binds directly at the promoter region of the p21 gene.
Depleting PRMT6 led to p21 induction, resulting in growth arrest. Importantly, this effect did not rely on p53, a master tumor suppressor that also interacts with p21. "Our data proves that by targeting PRMT6 we can re-activate the expression of the tumor suppressor p21 in cancer cells, which would lead to cell cycle arrest and would prevent tumor formation," says Guccione.
Chemists have developed a number of drugs that block PRMT6. However, many of these compounds also inhibit other members of the protein arginine methyltransferase (PRMT) family, resulting in intolerable side effects because of the broad-sweeping nature of these agents on cell physiology. Thus, "it would be very useful to find new inhibitors to specifically target PRMT6 activity," notes Guccione. Researchers in the United States recently discovered two compounds that specifically inhibit PRMT1. Meanwhile, the hunt for selective PRMT6-blockers continues.
Adding further weight to Guccione's team's findings, two research groups working in Europe independently confirmed that the p21 gene is a target of PRMT6 in human bone cancer cell lines. Intriguingly, one group found that p16 is also directly regulated by PRMT6, while the other linked p27 expression to PRMT6 activity. As a follow up, Guccione and his colleagues are now searching for other tumor suppressors affected by PRMT6. "We believe p21 is just one of many targets that are controlled directly by PRMT6," he says.
More information: Phalke, S., Mzoughi, S., Bezzi, M., Jennifer, N., Mok, W. C. et al. p53-Independent regulation of p21Waf1/Cip1 expression and senescence by PRMT6. Nucleic Acids Research 40, 9534–9542 (2012). dx.doi.org/10.1093/nar/gks858














